Edition: Hormones & Metabolism
15 March, 2026
In The News
Vol 1, Edition 10
How to Lower Cortisol Levels — Why Chronic Stress Is Making You Store Fat and What to Do About It
You’ve been told stress makes you eat more. That’s only part of the story. Cortisol directly instructs your body to store fat — specifically in the abdomen — independent of what you eat. Understanding the hormonal mechanism changes how you approach both stress and weight.

Published By: MAP30 Challenge
Authored By: John Shaw
74%
higher visceral fat in chronically stressed adults vs. low-stress peers
x4
greater insulin resistance risk with consistently elevated cortisol
6-8 hrs
of quality sleep reduces cortisol by up to 25% within one week
A common assumption — repeated in fitness culture and even in some medical offices — is that stress causes weight gain because stressed people eat more. The biology is more specific and more consequential than that. Cortisol, the body’s primary stress hormone, directly instructs fat cells to store energy in the abdominal region, suppresses insulin sensitivity, and elevates blood glucose — independent of how much you eat. Understanding this distinction is not academic. It changes which interventions actually work.
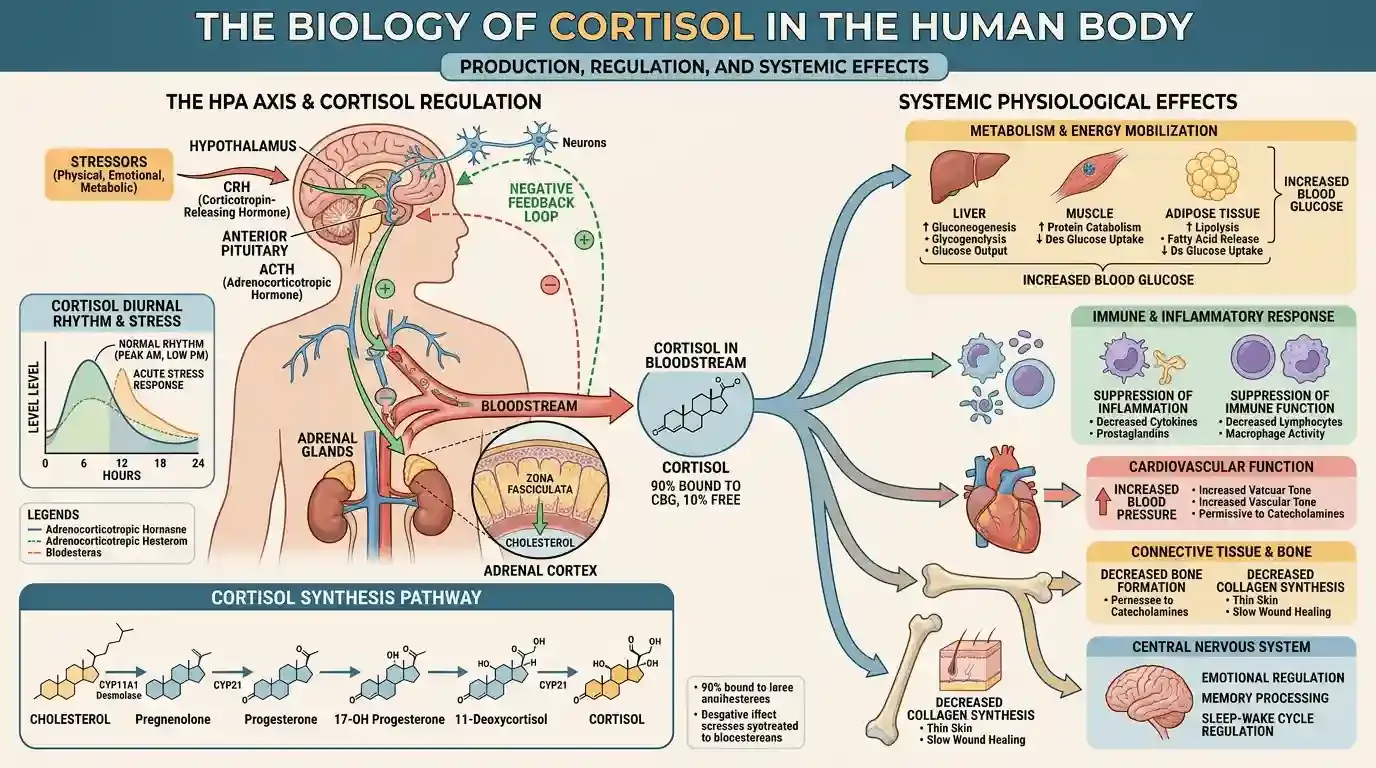
What Cortisol Actually Is — And Why It Exists
Cortisol is a glucocorticoid hormone produced by the adrenal glands — two small glands that sit atop your kidneys. It is released in response to physical or psychological stress, and in its acute form, it is one of the most important survival hormones in the human body.
When you encounter a genuine threat — a predator, a physical emergency — cortisol triggers a rapid cascade: blood glucose rises to fuel muscles, non-essential functions like digestion and reproduction are suppressed, the immune system is put on standby, and energy is mobilized for immediate use. This is the stress response working exactly as designed.
The problem is that the human stress system cannot distinguish between a charging predator and a difficult email. A contentious meeting, financial anxiety, poor sleep, or chronic undereating all trigger the same hormonal response. And when that response is activated repeatedly — day after day, week after week — cortisol stops being a survival tool and starts becoming a metabolic liability.
“Cortisol is not making you fat because it makes you hungry. It is making you fat because it is directly instructing your fat cells — specifically the ones in your abdomen — to store energy.”
The Direct Mechanism: How Cortisol Causes Belly Fat Storage
Most explanations of stress and weight gain focus on behavior: cortisol makes you crave high-calorie foods, cortisol disrupts sleep which increases hunger hormones, cortisol reduces willpower. All of this is true — but it misses the more fundamental mechanism.
Visceral adipose tissue — the deep abdominal fat that surrounds your organs — has a significantly higher density of cortisol receptors than subcutaneous fat elsewhere in the body. This is not incidental. It means visceral fat cells are far more sensitive to cortisol’s storage signals than fat cells in, say, your thighs or arms.
When cortisol is chronically elevated, it binds to these receptors and activates a process called adipogenesis — the differentiation of pre-adipocytes into mature fat cells. In practical terms: cortisol is not just filling existing fat cells. At sufficient levels, it is creating new ones, specifically in the visceral region.
CLINICAL STUDY — Bjørntorp P, Rosmond R. (2000)
"Obesity and cortisol." Nutrition, 16(10), 924–936.
Research by Bjørntorp and Rosmond at the University of Gothenburg demonstrated that chronically elevated cortisol preferentially drives fat deposition into visceral adipose tissue via glucocorticoid receptor activation. Subjects with higher cortisol reactivity showed significantly greater visceral fat accumulation independent of total caloric intake — establishing that the mechanism is hormonal, not purely behavioral.
What This Means: You can eat the same number of calories as someone with lower cortisol and accumulate more visceral fat. The variable is hormonal, not arithmetic.
Cortisol and Insulin Resistance: The Double Lock
The fat storage effect of cortisol is compounded by its relationship with insulin. Cortisol is physiologically antagonistic to insulin — meaning that when cortisol is elevated, it actively reduces insulin sensitivity in muscle and liver cells.
Here is the mechanism in sequence: cortisol elevates blood glucose by triggering gluconeogenesis in the liver (converting protein and fat stores into glucose). The pancreas responds by releasing more insulin to manage the elevated glucose. But cortisol simultaneously makes cells less responsive to that insulin. The result is chronically elevated both cortisol and insulin — a combination that essentially puts your metabolism in full storage mode.
Insulin’s primary role is to manage blood glucose — but when it is chronically elevated, it also suppresses lipolysis: the release of stored fatty acids for fuel. You cannot burn stored fat and have elevated insulin at the same time. Add chronically elevated cortisol on top, and you have a hormonal environment where fat storage is actively promoted and fat burning is actively blocked — regardless of how carefully you are eating.

Why This Matters for Weight Loss
If you are doing everything right — eating well, exercising, running a calorie deficit — but chronically stressed, your results will be blunted. Not because you lack discipline, but because elevated cortisol is working directly against the hormonal conditions needed for fat burning. Addressing stress is not a lifestyle accessory. For many people, it is the central metabolic lever.
What Chronic Cortisol Elevation Actually Looks Like

Most people think of cortisol as something you feel acutely — a racing heart before a presentation, the spike of adrenaline in an emergency. But chronic cortisol elevation is often subtler. It is the background hum of a life running at slightly too high a frequency, for too long.
The physical signs are recognizable once you know what to look for:
Accumulation of fat specifically in the abdomen and face — the “cortisol belly” pattern described in clinical literature
Energy crashes in the mid-afternoon, followed by a second wind late at night — consistent with disrupted cortisol rhythm
Difficulty falling asleep despite feeling exhausted — cortisol should be near its lowest at night; when it is not, sleep onset is compromised
Strong cravings for salt, sugar, and high-fat foods — driven by cortisol's suppression of leptin signaling, which impairs the brain's ability to register fullness and satisfaction
Slow recovery from exercise — elevated cortisol suppresses the anabolic hormones needed for muscle repair
Frequent illness — chronic cortisol suppresses immune function after an initial boost, leaving you more vulnerable to infection
The metabolic pattern that develops from chronic cortisol elevation — visceral fat accumulation, insulin resistance, and elevated triglycerides — is clinically indistinguishable from metabolic syndrome produced by a high-sugar, high-carbohydrate diet. Which is why the two conditions so frequently coexist and reinforce each other.
The Cortisol–Sleep–Weight Gain Loop
One of the most clinically important — and least discussed — cortisol mechanisms involves sleep. Cortisol follows a diurnal rhythm: it peaks in the early morning to help you wake up and ramps down throughout the day, reaching its lowest point around midnight. Chronic stress disrupts this rhythm, keeping evening cortisol elevated when it should be falling.
Elevated evening cortisol delays the release of melatonin, makes it harder to fall and stay asleep, and reduces the proportion of deep restorative sleep. The same disrupted sleep pattern also elevates ghrelin — the hunger hormone — which is why poor sleep and chronic stress produce persistent hunger that no amount of willpower resolves.
This matters for body composition in multiple ways: growth hormone — the primary hormone responsible for fat mobilization and muscle repair — is released almost exclusively during deep sleep. Poor sleep means less growth hormone, less fat burning, and slower recovery from exercise.
CLINICAL STUDY — Epel ES et al. (2000)
"Stress and body shape: Stress-induced cortisol secretion is consistently greater among women with central fat." Psychosomatic Medicine, 62(5), 623–632.
Epel and colleagues at UCSF found that women with greater visceral fat showed larger cortisol responses to standardized psychological stressors — and recovered more slowly afterward — than women with predominantly peripheral fat distribution. This created a self-reinforcing loop: visceral fat itself increases cortisol reactivity, which drives more visceral fat accumulation.
What This Means: The relationship between cortisol and visceral fat is bidirectional. Reducing stress reduces visceral fat — and reducing visceral fat also reduces cortisol reactivity. Both interventions accelerate each other.
How to Lower Cortisol Levels — What the Evidence Actually Supports
There is no shortage of advice about lowering cortisol online — some of it sound, much of it not. What follows is grounded in the clinical mechanisms described above, not general wellness rhetoric.
1. Sleep is the most powerful cortisol regulator available to you
Consistent sleep of seven to nine hours — not just duration but quality and timing — normalizes the cortisol diurnal rhythm faster than any other intervention. Research consistently shows that sleep restriction raises morning cortisol, while sleep restoration reduces it within days. The target is not just falling asleep but maintaining consistent sleep and wake times, reducing light exposure after dark, and keeping the bedroom cool.
2. Reduce chronic carbohydrate load — not just for insulin, but for cortisol
Blood glucose crashes — the kind produced by refined carbohydrates and added sugars — are themselves a cortisol trigger. The body interprets a sharp glucose drop as an emergency and releases cortisol to restore blood sugar via gluconeogenesis. Reducing refined carbohydrates eliminates this cycle and removes one of the most common sources of daily cortisol spikes that most people never identify as stress.
3. Zone 2 cardiovascular exercise — not high-intensity training
High-intensity exercise acutely raises cortisol. For someone already chronically stressed, adding intense daily training can worsen the hormonal picture rather than improve it. Low-to-moderate intensity exercise — walking, cycling at a conversational pace, easy swimming — reduces baseline cortisol over time without adding to the acute load. Thirty to forty-five minutes of Zone 2 cardio three to five times per week is the evidence-supported approach for cortisol management specifically.
4. Protein at breakfast — before caffeine if possible
Cortisol peaks in the morning and stimulates gluconeogenesis, which is why fasted blood glucose is often elevated in insulin-resistant individuals. Eating a protein-forward breakfast — eggs, meat, full-fat dairy — within an hour of waking blunts the morning cortisol spike and reduces subsequent glucose variability. Caffeine on an empty stomach amplifies the cortisol spike significantly; black coffee after a protein meal produces a substantially smaller cortisol response.
5. Deliberate downregulation — this is not optional
The nervous system does not downregulate cortisol automatically in a modern environment. It requires deliberate input. Research-supported interventions include diaphragmatic breathing (extending the exhale activates the parasympathetic nervous system), consistent exposure to natural light in the morning, and reducing screen and blue-light exposure in the two hours before bed. These are direct inputs into the autonomic nervous system that measurably reduce cortisol within sessions.
The Bottom Line on How to Lower Cortisol Levels
Cortisol does not respond to willpower or motivation. It responds to biological inputs: sleep quality, glucose stability, exercise intensity, and nervous system state. The most effective strategy addresses all four simultaneously — not because any single intervention is insufficient, but because cortisol dysregulation is rarely caused by one factor. The combination of consistent sleep, reduced refined carbohydrates, moderate exercise, and deliberate downregulation is the most evidence-supported protocol available without medication.
The Bottom Line on How to Lower Cortisol Levels

One of the most searched questions in metabolic health is how to tell if abdominal fat is primarily cortisol-driven or insulin-driven. In practice, both mechanisms are usually present and reinforce each other. But there are clinical patterns that can help distinguish the dominant driver.
Signs that cortisol is the dominant driver: fat accumulation in both the abdomen and face, significant energy dysregulation (wired at night, exhausted in the morning), elevated fasting blood glucose that fluctuates widely, and a history of chronic psychological or physical stress without proportionate dietary causes.
Signs that insulin resistance is the dominant driver: fat concentrated specifically in the abdomen without face involvement, strong sugar and carbohydrate cravings, postprandial energy crashes, elevated fasting insulin, and a dietary history heavy in refined carbohydrates and processed foods.
The honest clinical answer is that in most metabolically compromised adults, both are present. Cortisol drives insulin resistance; insulin resistance impairs cortisol metabolism. Addressing one helps the other. The MAP30 protocol addresses both simultaneously.
“Cortisol belly and insulin belly are not different problems with different solutions. They are two expressions of the same underlying metabolic disruption — and they respond to the same foundational interventions.”
Cortisol Belly vs. Insulin Resistance — How to Tell the Difference
One of the most searched questions in metabolic health is how to tell if abdominal fat is primarily cortisol-driven or insulin-driven. In practice, both mechanisms are usually present and reinforce each other. But there are clinical patterns that can help distinguish the dominant driver.
Signs that cortisol is the dominant driver: fat accumulation in both the abdomen and face, significant energy dysregulation (wired at night, exhausted in the morning), elevated fasting blood glucose that fluctuates widely, and a history of chronic psychological or physical stress without proportionate dietary causes.
Signs that insulin resistance is the dominant driver: fat concentrated specifically in the abdomen without face involvement, strong sugar and carbohydrate cravings, postprandial energy crashes, elevated fasting insulin, and a dietary history heavy in refined carbohydrates and processed foods.
The honest clinical answer is that in most metabolically compromised adults, both are present. Cortisol drives insulin resistance; insulin resistance impairs cortisol metabolism. Addressing one helps the other. The MAP30 protocol addresses both simultaneously.
What This Means for the MAP30 Challenge
The MAP30 Challenge is built around the metabolic principles described in this article. The 30-day protocol is designed to address the hormonal environment — not just caloric intake — that determines whether your body stores fat or burns it.
The dietary changes in MAP30 directly reduce the blood glucose variability that drives daily cortisol spikes. The sleep protocols in MAP30 are designed to normalize the cortisol diurnal rhythm. The movement framework in MAP30 is calibrated to reduce baseline cortisol rather than add to it.
If you have been doing the right things and not seeing results, cortisol dysregulation may be the variable you have not yet addressed. The research is clear: without managing the hormonal environment, even a well-designed diet and exercise program produces blunted results. MAP30 addresses the environment first.
Latest Articles

What Is Insulin Resistance
You don't need a diabetes diagnosis to have insulin resistance. Millions of Americans are living with...

Calorie Deficit Diets?
The calorie deficit model has been the foundation of weight loss advice for over a century. But millions of people...
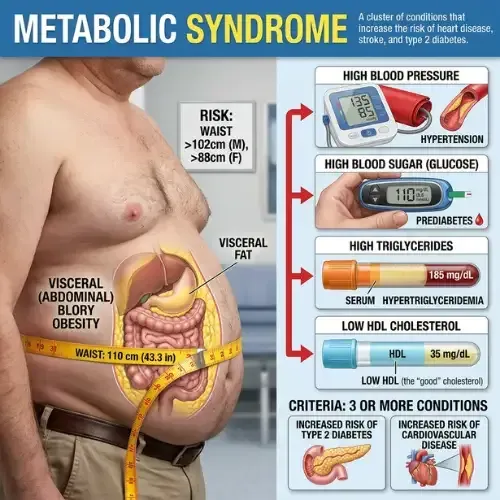
Metabolic Syndrome
Most people who have metabolic syndrome don’t know it. There’s no single dramatic symptom...

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
Disclaimer: The information in this article is for educational and historical context only and does not constitute medical or dietary advice. Nutritional science is an evolving field and individual dietary needs vary. Consult a qualified healthcare provider or registered dietitian before making significant changes to your diet or health management plan.
FAQ's
1. Does stress cause weight gain?
Yes — through a hormonal mechanism, not just behavioral changes. Chronic stress elevates cortisol, which directly promotes visceral fat storage by activating glucocorticoid receptors in abdominal adipose tissue. Cortisol also raises blood glucose and insulin, creating a hormonal environment that suppresses fat burning and promotes fat storage independent of caloric intake. Research by Epel et al. (2000) confirmed that women with greater cortisol reactivity to stress showed significantly more visceral fat accumulation.
2. What are the symptoms of high cortisol?
Common symptoms of chronically elevated cortisol include: visceral fat accumulation (especially around the abdomen), difficulty losing weight despite dietary changes, fatigue combined with difficulty sleeping, carbohydrate and sugar cravings, elevated fasting blood glucose, high blood pressure, poor recovery from exercise, brain fog, and low mood or anxiety. These symptoms overlap significantly with insulin resistance because chronic cortisol elevation drives insulin elevation.
3. How does cortisol cause belly fat?
Cortisol promotes visceral fat storage through two mechanisms. First, it activates glucocorticoid receptors in abdominal adipose tissue, directly promoting fat storage in the belly region. Second, cortisol raises blood glucose by stimulating gluconeogenesis in the liver, which raises insulin, which further promotes fat storage and suppresses lipolysis. Visceral fat itself produces inflammatory cytokines that worsen insulin resistance, creating a self-reinforcing cycle.
4. How do you lower cortisol levels naturally?
The most evidence-supported interventions for lowering cortisol naturally are: consistent quality sleep of 7–9 hours (sleep deprivation is one of the most potent cortisol elevators); Zone 2 aerobic exercise such as long walks, which reduces baseline cortisol without adding physiological stress; carbohydrate stabilization to eliminate blood sugar swings that trigger cortisol spikes; diaphragmatic breathing with extended exhales to activate the parasympathetic nervous system; and visceral fat reduction, which reduces the inflammatory load that sustains HPA axis activation.
5. What is the connection between cortisol and insulin resistance?
Cortisol and insulin resistance are directly connected. Chronically elevated cortisol raises blood glucose through hepatic gluconeogenesis, which requires elevated insulin to manage. Over time, this sustained insulin elevation drives cellular insulin resistance. Additionally, cortisol directly impairs insulin signaling at the cellular level by interfering with GLUT4 transporter expression in muscle cells. The result is a bidirectional relationship: chronic stress worsens insulin resistance, and insulin resistance amplifies the metabolic consequences of cortisol elevation.
6. Does poor sleep raise cortisol?
Yes. Sleep is one of the primary regulators of the cortisol diurnal rhythm. Cortisol should peak in the early morning to support waking and gradually decline through the day. Sleep deprivation disrupts this rhythm, elevating evening and nighttime cortisol when it should be low. Even a single night of poor sleep measurably increases insulin resistance the following morning. Chronic poor sleep is an independent driver of visceral fat accumulation, elevated fasting insulin, and metabolic syndrome risk.
Sources
Björntorp P — "Do stress reactions cause abdominal obesity and comorbidities?" (Obesity Reviews, 2001)
https://pubmed.ncbi.nlm.nih.gov/12119665/
Epel ES et al. — "Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat" (Psychosomatic Medicine, 2000)
https://pubmed.ncbi.nlm.nih.gov/11020091/
Leproult R & Van Cauter E — "Effect of 1 week of sleep restriction on testosterone levels in young healthy men" (JAMA, 2011)
https://pubmed.ncbi.nlm.nih.gov/21632481/
Dallman MF et al. — "Chronic stress and obesity: a new view of comfort food" (PNAS, 2003)
https://pubmed.ncbi.nlm.nih.gov/12975485/
Rosmond R — "Role of stress in the pathogenesis of the metabolic syndrome" (Psychoneuroendocrinology, 2005)

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
