Edition: Metabolic Disease
15 March, 2026
In The News
Vol 1, Edition 11
Ozempic and GLP-1: What the Weight Loss Drug Craze Isn't Telling You
GLP-1 drugs work. That part is true. What the headlines skip is how they work, what happens when you stop, and what your body can do naturally with the same hormonal pathway — without the injections, the cost, or the side effects.

Published By: MAP30 Challenge
Authored By: John Shaw
Ozempic is not a mystery drug. It is a pharmaceutical replica of a hormone your body already makes.
GLP-1 — glucagon-like peptide-1 — is a gut hormone produced naturally after eating. It signals the pancreas to release insulin, tells the brain you're full, slows gastric emptying, and suppresses glucagon — the hormone that raises blood sugar. When it works properly, it is one of the most powerful metabolic regulators in the body.
In people with type 2 diabetes and severe obesity, GLP-1 signaling is impaired. The hormone is produced but the response is blunted. Ozempic — semaglutide — is a synthetic GLP-1 receptor agonist that activates the same receptors with far greater potency and duration than the natural hormone. It works because the underlying biology is real.
What the weight loss drug conversation largely skips is the full picture: what these drugs are actually doing, what the research shows about long-term outcomes, what happens when you stop, and — critically — what the natural GLP-1 pathway looks like when the metabolic environment is working as designed.
"Ozempic doesn't introduce a new biological mechanism. It hijacks an existing one — and amplifies it to a level the body was never designed to sustain indefinitely."
Ozempic vs. Natural GLP-1 Activation: Same Outcome, Different Path
💉 Ozempic / Semaglutide
Pharmaceutical GLP-1 receptor agonist
Mechanism
Synthetic compound activates GLP-1 receptors at far higher potency than the natural hormone. Half-life: ~1 week.
Appetite suppression
Strong and consistent — achieved by pharmacologically overriding hunger signals in the hypothalamus.
Weight loss
15–22% mean body weight in clinical trials. Real and significant outcomes.
Underlying cause
Not addressed. Insulin resistance, gut dysbiosis, and leptin resistance persist during treatment.
When stopped
~67% of lost weight regained within 1 year. Metabolic markers return to baseline (STEP 4 trial).
Cost
$900–$1,400/month without insurance. Requires ongoing prescription and administration.
🌿 Natural GLP-1 Activation
Restoring the body's own GLP-1 system
Mechanism
Supports L-cell GLP-1 secretion and receptor sensitivity through diet, gut health, and fasting.
Appetite suppression
Gradual — achieved by restoring the hormonal environment that allows natural satiety signaling to function.
Weight loss
Slower onset but durable — driven by hormonal normalization rather than pharmacological override.
Underlying cause
Directly addressed. Insulin resistance, gut health, and leptin sensitivity all improve with the metabolic environment.
When stopped
N/A — outcomes persist because the underlying metabolic dysfunction has been addressed, not bypassed.
Cost
Dietary and lifestyle change. No prescription, no injection, no ongoing pharmaceutical cost.
What GLP-1 Actually Does — The Biology Behind the Drug
GLP-1 is secreted by L-cells in the small intestine and colon in response to food — particularly protein and fat. Its effects are rapid, potent, and multiple:
• Stimulates insulin secretion from pancreatic beta cells in a glucose-dependent manner — insulin rises only when blood glucose is elevated, which is why GLP-1 drugs don't typically cause hypoglycemia in non-diabetic patients
• Suppresses glucagon — the liver hormone that raises blood glucose — reducing the liver's glucose output between meals
• Slows gastric emptying — food moves more slowly from the stomach into the small intestine, reducing the rate of glucose absorption and extending the feeling of fullness
• Acts on the hypothalamus to suppress appetite — GLP-1 receptors in the brain receive the hormone's signal directly and reduce the drive to eat
• Reduces inflammation — GLP-1 receptors are present on immune cells and their activation has been shown to reduce inflammatory markers
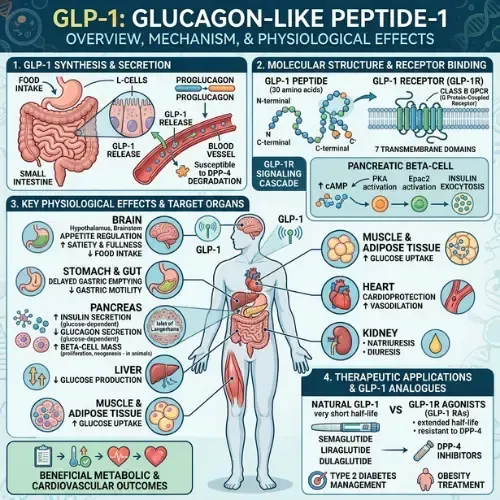
In a metabolically healthy person, this system works continuously and automatically. After eating, GLP-1 rises, insulin is appropriately stimulated, the meal is processed efficiently, and appetite is suppressed until the next meal.
In insulin resistance and type 2 diabetes, several things go wrong simultaneously: GLP-1 secretion is reduced after meals, GLP-1 receptor sensitivity is impaired, and the incretin effect — the amplification of insulin release triggered by gut hormones — is substantially diminished. The body's natural GLP-1 system is there, but underperforming.
What Ozempic and GLP-1 Drugs Actually Do
Semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro) are synthetic compounds that activate GLP-1 receptors with far greater potency and duration than the natural hormone. Natural GLP-1 has a half-life of approximately two minutes — it is rapidly degraded by the enzyme DPP-4. Semaglutide is engineered to resist this degradation, giving it a half-life of approximately one week with a single weekly injection.
The effect is a sustained, high-level activation of GLP-1 receptors that produces consistent appetite suppression, reduced gastric emptying, and improved insulin secretion throughout the week. The clinical trial results are real: the STEP trials showed average weight loss of 15–17% of body weight in people with obesity over 68 weeks with semaglutide. The SURMOUNT trials with tirzepatide showed up to 22% weight loss.
These are significant outcomes. For people with severe obesity and related metabolic disease, GLP-1 drugs represent a meaningful clinical intervention. That is not the debate.
🔬 CLINICAL STUDY
Wilding JPH et al. (2021)
STEP 1 Trial
New England Journal of Medicine, 384(11), 989–1002.
A randomized controlled trial of 1,961 adults with obesity found that once-weekly semaglutide 2.4mg produced mean weight loss of 14.9% over 68 weeks compared to 2.4% with placebo. Significant improvements in cardiometabolic risk factors were observed including blood pressure, triglycerides, and fasting glucose.
What This Means: GLP-1 receptor agonists produce clinically significant weight loss and metabolic improvement in people with obesity. The effect size is real and substantially larger than diet and exercise interventions in trial settings. The critical question — what happens after the drug is stopped — is addressed in the withdrawal data below.
The Part Nobody Is Talking About: What Happens When You Stop
The most important clinical data on GLP-1 drugs is not in the weight loss headlines. It is in the withdrawal studies.
The STEP 4 trial followed participants who had achieved significant weight loss on semaglutide and then either continued the drug or switched to placebo. Within one year of stopping, participants in the withdrawal group regained approximately two-thirds of their lost weight. Most metabolic markers — blood pressure, blood glucose, lipids — returned toward baseline.
The mechanism is straightforward. GLP-1 drugs suppress appetite through pharmacological activation of GLP-1 receptors. They do not address the underlying metabolic dysfunction — insulin resistance, leptin resistance, gut microbiome disruption — that drove the obesity and metabolic disease in the first place. When the drug is stopped, those underlying conditions remain. Appetite returns. The hormonal environment that promotes fat storage reasserts itself.
The Appetite Rebound Mechanism:
GLP-1 drugs suppress appetite partly by reducing the activity of AgRP neurons in the hypothalamus — neurons that drive hunger. Research published in Nature (2024) found that after stopping GLP-1 drugs, AgRP neuron activity rebounds significantly, producing hunger levels that exceed pre-treatment baseline in some patients. The drug suppresses the hunger signal. It does not fix the system generating it.
The Biggest Misconception About GLP-1 Medicines
The most widespread misconception about GLP-1 drugs is that they are treating the underlying cause of obesity and metabolic disease. They are not. They are treating a symptom — excess weight and elevated blood glucose — by pharmacologically overriding the appetite and insulin systems that have become dysfunctional.
This matters because it defines what happens after the drug. If the underlying metabolic dysfunction — insulin resistance, gut dysbiosis, chronic inflammation, leptin resistance — is not addressed during the period of drug-induced weight loss, the body simply returns to its pre-treatment state when the drug is removed.
The drug creates a window. What you do with that window determines whether the outcomes last. For many patients, that window is never used to address the metabolic environment that caused the problem — because neither the prescribing physician nor the patient understands that the drug is not doing that work.

🔬 CLINICAL STUDY
Knop FK et al. (2023)
STEP 4 Withdrawal
Diabetes, Obesity and Metabolism, 25(3), 656–667.
Analysis of the STEP 4 trial found that participants who discontinued semaglutide after 20 weeks of treatment regained a mean of 6.9% of body weight within the following 48 weeks, compared to continued weight loss of -7.9% in those who maintained treatment. Cardiometabolic markers including blood pressure, glucose, and lipids all returned toward baseline in the withdrawal group.
⚠️ What This Means: GLP-1 drug efficacy is contingent on continued administration. Without addressing the underlying metabolic dysfunction during the treatment period, discontinuation produces near-complete reversal of outcomes within one year.
How to Increase GLP-1 Naturally — What the Research Shows
Protein at meals
Protein is the most potent natural stimulator of GLP-1 secretion. High-protein meals produce significantly larger GLP-1 responses than high-carbohydrate meals at equivalent caloric content. Research consistently shows that prioritizing protein at breakfast — the meal most commonly skipped or replaced with refined carbohydrates — produces the strongest and most sustained GLP-1 response of the day.
Dietary fiber and the microbiome
Short-chain fatty acids produced by gut bacteria fermenting dietary fiber — particularly butyrate and propionate — directly stimulate L-cells to secrete GLP-1. A healthy, diverse gut microbiome producing adequate SCFAs is one of the most reliable predictors of robust natural GLP-1 response. Ultra-processed food consumption, which degrades microbiome diversity, directly reduces GLP-1 output through this pathway.
Fasting windows
Intermittent fasting improves GLP-1 receptor sensitivity over time. Research on time-restricted eating consistently shows enhanced GLP-1 response after the same meal following a fasting period compared to continuous eating patterns. The mechanism involves upregulation of GLP-1 receptor expression in both gut tissue and the hypothalamus.

Reducing ultra-processed food
Ultra-processed foods produce a blunted GLP-1 response compared to whole food meals of equivalent caloric content. The rapid digestion of refined carbohydrates bypasses the portion of the small intestine where L-cells are most concentrated, reducing the GLP-1 signal that would normally accompany a meal of similar energy content.
Who GLP-1 Drugs Are Actually Appropriate For
This article is not an argument against GLP-1 drugs for every patient. For people with severe obesity, type 2 diabetes with significant cardiovascular risk, or metabolic disease that has not responded to lifestyle intervention, GLP-1 drugs represent a legitimate medical tool — one with a better evidence base than most weight loss interventions that preceded them.
The concern is the population the drugs are being marketed to and prescribed for: people with moderate weight issues, people who have not attempted metabolic dietary change, people using the drug as an alternative to addressing the underlying hormonal and gut-related dysfunction that is actually driving their symptoms. For this population, the drug treats the number on the scale. It does not teach the body to regulate itself.
The healthcare crisis that GLP-1 drugs may produce is not a crisis of side effects — though those exist and include pancreatitis risk, thyroid concerns, and the well-documented muscle loss that accompanies rapid weight reduction without adequate protein. The crisis is a generation of people who lose weight, stop the drug, regain the weight, and never understood that what they actually needed was metabolic rehabilitation.
What This Means for the MAP30 Challenge
MAP30 is built around the metabolic conditions that allow the body's own GLP-1 system to work as designed. The dietary framework — reducing refined carbohydrates, prioritizing protein, eliminating ultra-processed foods — directly supports GLP-1 secretion and receptor sensitivity. The gut health framework supports the microbiome-SCFA-L-cell pathway that is the most consistent natural driver of GLP-1 output. The fasting protocols improve receptor upregulation over time.
The goal is not to replace a drug. The goal is to make the drug unnecessary — by restoring the metabolic environment in which the body's own regulatory systems can function as they were designed to.
This article is part of our Metabolic Disease series.
Link Here: Metabolic Disease
Latest Articles

What Is Insulin Resistance
You don't need a diabetes diagnosis to have insulin resistance. Millions of Americans are living with...

Calorie Deficit Diets?
The calorie deficit model has been the foundation of weight loss advice for over a century. But millions of people...
Metabolic Syndrome
Most people who have metabolic syndrome don’t know it. There’s no single dramatic symptom...

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
Disclaimer: The information in this article is for educational and historical context only and does not constitute medical or dietary advice. Nutritional science is an evolving field and individual dietary needs vary. Consult a qualified healthcare provider or registered dietitian before making significant changes to your diet or health management plan.
FAQ's
1. What is GLP-1 and how does it work?
GLP-1 (glucagon-like peptide-1) is a gut hormone produced after eating that stimulates insulin secretion, suppresses glucagon, slows gastric emptying, and signals the brain to reduce appetite. It is one of the primary hormonal regulators of blood sugar and satiety. GLP-1 drugs like Ozempic are synthetic versions that activate the same receptors with far greater potency and duration than the natural hormone.
2. What is the biggest misconception about GLP-1 medicines?
The biggest misconception is that GLP-1 drugs treat the underlying cause of obesity or metabolic disease. They do not. They pharmacologically override the appetite and insulin systems that have become dysfunctional — producing weight loss while the underlying insulin resistance, gut dysbiosis, leptin resistance, and hormonal disruption remain unaddressed. When the drug stops, those conditions reassert themselves and weight typically returns.
3. What happens when you stop taking Ozempic or GLP-1 drugs?
Clinical data from the STEP 4 trial and subsequent research consistently shows that stopping GLP-1 drugs results in significant weight regain — approximately two-thirds of lost weight within one year in most patients. Cardiometabolic markers including blood pressure, blood glucose, and lipids return toward pre-treatment levels. Appetite rebounds, in some cases above pre-treatment baseline, due to rebound activation of hunger-driving neurons in the hypothalamus.
4. How do you increase GLP-1 naturally?
GLP-1 secretion and receptor sensitivity can be improved through: high-protein meals, which are the most potent natural stimulators of GLP-1 secretion; dietary fiber diversity, which feeds gut bacteria that produce SCFAs stimulating L-cell GLP-1 output; fasting windows, which improve GLP-1 receptor sensitivity and expression; and eliminating ultra-processed foods, which produce blunted GLP-1 responses compared to whole food meals.
5. Is Ozempic appropriate for everyone who wants to lose weight?
GLP-1 drugs have a legitimate evidence base for people with severe obesity, type 2 diabetes, or significant cardiovascular metabolic risk. The concern is their use as a first-line intervention for people who have not addressed underlying metabolic dysfunction through dietary change. For this population, the drug treats the symptom without rehabilitating the system — making long-term outcomes contingent on indefinite drug use.
6. Does Ozempic cause muscle loss?
Yes — this is one of the most documented and least discussed side effects of rapid weight loss on GLP-1 drugs. Clinical trials show that a significant proportion of weight lost on GLP-1 drugs — estimates range from 25–40% — is lean mass rather than fat. Muscle loss reduces resting metabolic rate, impairs insulin sensitivity in skeletal muscle (one of the primary sites of glucose disposal), and increases the likelihood of weight regain after discontinuation. Adequate protein intake and resistance exercise during treatment are critical to mitigating this effect.
Sources
Wilding JPH et al. — 'Once-Weekly Semaglutide in Adults with Overweight or Obesity' STEP 1 Trial (NEJM, 2021)
https://pubmed.ncbi.nlm.nih.gov/33567185/
Knop FK et al. — 'Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes' / STEP 4 withdrawal analysis (Diabetes, Obesity and Metabolism, 2023)
https://pubmed.ncbi.nlm.nih.gov/36461087/
Drucker DJ — 'GLP-1 physiology informs the pharmacotherapy of obesity' (Molecular Metabolism, 2022)
https://pubmed.ncbi.nlm.nih.gov/34626818/
Holst JJ — 'The physiology of glucagon-like peptide 1' (Physiological Reviews, 2007)
https://pubmed.ncbi.nlm.nih.gov/17928588/
Jastreboff AM et al. — SURMOUNT-1 Trial: Tirzepatide once weekly for treatment of obesity (NEJM, 2022)
https://pubmed.ncbi.nlm.nih.gov/35658024/
Müller TD et al. — 'Glucagon-like peptide 1 (GLP-1)' (Molecular Metabolism, 2019)

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
