12 April, 2026
Edition: Hormones & Metabolism
Vol 1, Edition 27
Is Sleep Slowing Your Metabolism? The Hormonal Connection Most People Miss
You're doing everything right — eating well, staying active, managing stress as best you can. And yet the scale won't move, your energy is flat, and you're hungry in a way that feels out of proportion to what you've eaten. The variable you haven't accounted for may be happening every night while you're unconscious.

Published By: MAP30 Challenge | Authored By: John Shaw
Article Directory: https://map30challenge.com/article-directory
Sleep is not a passive state. While you are unconscious, your body is running one of its most metabolically active and hormonally complex processes of the entire day. Growth hormone is released. Cortisol is reset. Leptin rises to signal adequate energy stores. Insulin sensitivity is restored in preparation for the morning. The entire hormonal orchestra that governs hunger, fat burning, and metabolic rate is being recalibrated during the hours you spend asleep.
When sleep is shortened, fragmented, or chronically poor in quality, this recalibration is disrupted. Not partially — substantially. A single night of poor sleep produces measurable increases in cortisol, measurable drops in leptin, measurable rises in ghrelin, and measurable reductions in insulin sensitivity the following morning. These are not minor fluctuations. They are hormonal shifts significant enough to produce real changes in hunger, fat storage, and metabolic rate — and they compound with every subsequent night of poor sleep.
Most metabolic health conversations focus on what you eat and when you eat it. Sleep rarely makes the list. This is a significant oversight — because no dietary intervention, no fasting protocol, and no exercise program fully compensates for the hormonal disruption that chronic sleep restriction produces. Sleep is not a lifestyle accessory to metabolic health. It is a biological requirement for it.
The Cortisol Reset — Why Sleep Timing Matters as Much as Duration
Cortisol follows a diurnal rhythm — it peaks sharply in the early morning to wake you up and ramp your body into activity, then declines steadily through the day, reaching its lowest point around midnight. This rhythm is not incidental. It is the hormonal clock that coordinates energy availability, inflammation, and metabolic rate across the entire 24-hour cycle.
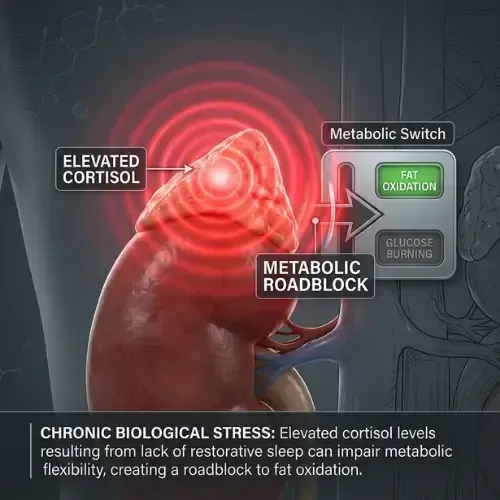
Chronic sleep restriction disrupts this rhythm in two critical ways. First, it elevates evening cortisol — the period when cortisol should be at its lowest. Elevated evening cortisol delays melatonin release, makes sleep onset harder, and reduces the proportion of deep slow-wave sleep even when total sleep duration appears adequate. Second, it elevates morning cortisol beyond its normal peak — producing a hyperactivated stress response at the start of the day that drives gluconeogenesis, raises fasting blood glucose, and promotes visceral fat accumulation.
Research from the University of Chicago found that sleep restriction to four hours per night for six nights produced cortisol levels in the afternoon and evening that were significantly higher than after normal sleep — a pattern associated with accelerated aging, increased visceral fat deposition, and impaired glucose tolerance. (Spiegel K et al. — Lancet, 1999)
As covered in the Cortisol and Weight Gain article in this library, visceral adipose tissue has a high density of glucocorticoid receptors — making it specifically sensitive to cortisol's fat storage signal. Every night of elevated evening cortisol is a night of preferential visceral fat deposition. This is why the relationship between poor sleep and abdominal weight gain is not anecdotal. It is receptor-mediated and hormonally direct.
What Happens to Your Metabolic Hormones While You Sleep
What Happens to Your Metabolic Hormones While You Sleep
7–9 hours of quality sleep vs. 5–6 hours of poor or fragmented sleep
✅ 7–9 Hours Quality Sleep
The overnight hormonal recalibration completes
Cortisol — falls to nadir
Reaches its lowest point around midnight. Diurnal rhythm intact. Morning cortisol peaks appropriately to wake you without driving fat storage.
Growth Hormone — pulses strongly
70–80% of daily GH secretion occurs during deep slow-wave sleep in the first 2–3 hours. Fat mobilization and lean mass repair running at full capacity.
Leptin — rises overnight
Signals the brain that energy stores are adequate. Appetite is suppressed through the overnight fast. You wake without urgent hunger.
Ghrelin — appropriately suppressed
Hunger hormone is kept low overnight. Morning appetite is manageable rather than urgent and carbohydrate-driven.
Insulin sensitivity — restored
Cellular glucose transporters reset. Inflammatory markers normalized. You begin the day with full insulin sensitivity.
⚠️ 5–6 Hours Poor Sleep
The recalibration is interrupted — every night
Cortisol — stays elevated
Evening cortisol remains high instead of falling. Drives visceral fat deposition overnight. Morning cortisol spikes beyond normal — elevating fasting glucose.
Growth Hormone — suppressed
Deep slow-wave sleep is shortened or fragmented. GH pulses are reduced or absent. Fat mobilization and tissue repair are compromised every night.
Leptin — drops 18–24%
Brain receives inadequate satiety signal. Perceived energy deficit despite full fat stores. Drives hunger and food-seeking behavior from the moment you wake.
Ghrelin — rises 18–28%
Hunger hormone elevated. Appetite for high-calorie, high-carbohydrate foods specifically increases by up to 45%. This is physiological — not a willpower failure.
Insulin sensitivity — reduced 25%
Overnight restoration incomplete. You begin the day already insulin resistant. Equivalent metabolic impact to six months of high-fat dietary disruption.
Growth Hormone — The Fat-Burning Window You're Sleeping Through
Growth hormone is the primary hormone responsible for fat mobilization, lean mass preservation, and cellular repair. In adults, approximately 70 to 80% of daily growth hormone secretion occurs during sleep — specifically during the first two to three hours of sleep in the deepest stages of slow-wave sleep.
Growth hormone's metabolic role is the functional opposite of insulin. Where insulin drives fat storage, growth hormone drives fat mobilization — directly stimulating lipolysis in adipose tissue and promoting the use of fat as fuel. Where insulin suppresses protein breakdown, growth hormone supports muscle protein synthesis. The overnight period, when insulin is low and growth hormone is pulsing through slow-wave sleep cycles, is the body's primary daily fat-burning and tissue repair window.
Poor sleep directly reduces growth hormone secretion. Sleep fragmentation, shortened sleep duration, and elevated evening cortisol all suppress the slow-wave sleep stages during which growth hormone is released. People who consistently sleep six hours or less show significantly reduced nocturnal growth hormone pulses compared to those sleeping seven to nine hours. The fat-burning window that should occur every night is shortened or eliminated — and no supplement, dietary intervention, or daytime activity replicates the growth hormone release that deep sleep produces.
This compounds with age. Growth hormone secretion naturally declines from the third decade of life onward. Chronic sleep restriction accelerates this decline. For adults over 40 already experiencing reduced growth hormone output, poor sleep removes one of the last reliable daily stimuli for fat mobilization and lean mass preservation. The combination of age-related decline and sleep-driven suppression is a significant contributor to the metabolic changes most people attribute to aging alone.
"You cannot out-diet poor sleep. The hormonal damage that two weeks of six-hour nights produces is measurable on a blood test — and no amount of clean eating reverses it while the sleep deprivation continues."
Leptin and Ghrelin — How Sleep Controls Your Hunger Hormones
Of all the hormonal consequences of poor sleep, the disruption of hunger signaling is perhaps the most immediately felt — and the most directly connected to weight gain.
Leptin and ghrelin work in opposition. Leptin — produced by fat cells — signals the brain that energy stores are adequate and suppresses appetite. Ghrelin — produced by the stomach — signals hunger and drives food-seeking behavior. In healthy sleep, leptin rises overnight to maintain satiety through the fast, and ghrelin is suppressed. The result is that most people wake up not particularly hungry for the first hour or two of the morning.
A landmark study by Spiegel and colleagues found that restricting sleep to two nights of four hours produced a 24% reduction in leptin and an 18% increase in ghrelin compared to two nights of ten hours. Hunger ratings increased by 24% — and appetite for high-calorie, high-carbohydrate foods specifically increased by 45%. (Spiegel K et al. — PLOS Medicine, 2004)
This is not a subtle effect. A 24% drop in leptin and an 18% rise in ghrelin represents a hormonal environment that produces genuine, physiological hunger — not psychological craving or habitual eating. The person experiencing this does not lack willpower. Their hunger hormones have been disrupted by insufficient sleep, and the biological drive to eat — particularly high-calorie, high-carbohydrate foods — is being generated by that disruption, not by any deficit of self-control.

The increase in appetite for refined carbohydrates specifically is not accidental. Ghrelin activates reward centers in the brain that are particularly responsive to high-glycemic foods — creating a hormonal pull toward exactly the foods that will produce the largest insulin spike and the most visceral fat storage. Poor sleep does not just make you hungry. It makes you hungry for the worst possible foods in a metabolic context.
Insulin Sensitivity — The Metabolic Reset That Only Happens During Sleep
Sleep is when insulin sensitivity is restored. During the overnight fast, as insulin falls to its basal level and the growth hormone window operates, cells in the liver and skeletal muscle restore their responsiveness to insulin's signal. This nightly reset is why insulin sensitivity in healthy adults is at its best in the morning — before eating has begun and after a full night of sleep has completed the restoration process.
One Night of Poor Sleep
reduces insulin sensitivity by up to 25% the following morning — equivalent to the metabolic impact of six months of a high-fat diet
Donga E et al. — Journal of Clinical Endocrinology & Metabolism, 2010
For people already managing insulin resistance or prediabetes, this means that two weeks of consistently poor sleep can measurably worsen fasting insulin and HOMA-IR even if the dietary and fasting protocol remains unchanged. Sleep is not a supplement to metabolic health interventions. It is the foundation they rest on.
I got the most direct personal experience of this during a seven-day water fast. By day four, I slept nine hours and woke up feeling like I hadn't felt in decades. No grogginess, no reaching for coffee just to function, no mid-morning crash at 10am. What I realized afterward is that I had simply forgotten what it felt like to wake up with a fully rested brain running on clean fuel — and what a night of genuinely restored insulin sensitivity and a full growth hormone window actually feels like in the morning. Most of us have been sleeping short or poorly for so long we've accepted the tired version of ourselves as normal. It isn't normal. It's the metabolic consequence of not protecting the overnight window. — John Shaw
How Much Sleep Is Actually Required — What the Research Shows
The research on sleep duration and metabolic health is remarkably consistent. Seven to nine hours of quality sleep per night is the range associated with optimal hormonal function, normal insulin sensitivity, and appropriate leptin and ghrelin balance. Below seven hours, measurable metabolic impairment begins. Below six hours, the impairment is substantial and compounds rapidly with each successive night.
The quality dimension is as important as the duration. Six hours of fragmented, light sleep produces worse metabolic outcomes than five and a half hours of consolidated, deep sleep — because the growth hormone pulses and cortisol normalization that occur during deep slow-wave sleep are disrupted by fragmentation even when total time in bed is adequate. The conditions for deep sleep — consistent sleep and wake times, dark and cool sleeping environment, minimal light and screen exposure before bed — are not optional lifestyle preferences. They are the environmental inputs that allow the overnight hormonal recalibration to run to completion.
Signs That Poor Sleep May Be Affecting Your Metabolism
The signals tend to appear in combination — one rarely shows up alone. If you're checking off three or more of these, sleep is likely part of the metabolic picture:
Waking up hungry — leptin has not risen adequately overnight to suppress appetite through the fast
Strong carbohydrate cravings first thing in the morning — ghrelin elevation driving appetite for high-glycemic foods before the day has started
Mid-afternoon energy crash — cortisol rhythm disruption producing the characteristic late-day energy drop regardless of what you ate at lunch
Difficulty losing weight despite doing everything else right — insulin resistance from sleep restriction quietly offsetting the dietary improvements you're making
Fasting blood glucose higher than usual — cortisol-driven gluconeogenesis elevating morning glucose before you've eaten a single thing
Elevated fasting insulin on blood tests — overnight insulin sensitivity restoration was incomplete — the reset didn't finish
Wired at night, exhausted in the morning — the classic disrupted cortisol diurnal rhythm, running backwards from how it should
What Actually Improves Sleep Quality — The Metabolic Approach
Most sleep advice starts and ends with sleep hygiene — screens, timing, temperature, darkness. Those things matter and are worth implementing. But there is a metabolic dimension to sleep quality that almost nobody talks about, and for anyone managing insulin resistance or working to restore metabolic health it is the most directly actionable lever of all.
It starts with what you eat in the evening and when you stop eating it. Blood glucose fluctuations overnight — produced by a high-glycemic evening meal — activate the cortisol response as the body manages the glucose crash that follows the spike. This disrupts sleep architecture, reduces slow-wave sleep, and elevates morning cortisol before you've even opened your eyes. A low-glycemic evening meal — protein and fat forward, minimal refined carbohydrates — produces stable overnight blood glucose, which keeps cortisol appropriately low and gives sleep architecture the conditions it needs. Then the timing of the last meal compounds this: eating large meals close to bedtime keeps insulin elevated during the early sleep period, directly suppressing growth hormone release during the precise window when it should be pulsing. The MAP30 eating window ending several hours before sleep is not just about fasting duration — it is about letting insulin fall far enough before sleep onset that the growth hormone window opens properly.

From there, evening cortisol is the next lever. As covered in the Cortisol article in this library, deliberate downregulation — consistent wind-down routines, reduced blue light, diaphragmatic breathing, stable sleep timing — directly lowers evening cortisol and protects sleep architecture. For people whose poor sleep is cortisol-driven rather than glucose-driven, this is the intervention, not a sleep supplement.
What most people miss is that the relationship runs both ways. Insulin resistance doesn't just result from poor sleep — it contributes to it. Elevated inflammatory markers, disrupted overnight glucose regulation, and impaired cellular repair all feed back into sleep quality. As insulin resistance improves through dietary change, sleep frequently improves as a secondary consequence — not because the sleep problem was directly targeted, but because the metabolic environment that was disrupting it has been corrected. The sleep gets better because the system running underneath it has been fixed. Seven to nine hours is not a recommendation. For metabolic recovery, it is a requirement.
This article is part of our Hormones & Metabolism series. Hormones & Metabolism
Latest Articles
Intermittent Fasting Help With Insulin Resistance
Intermittent fasting is the most talked-about dietary intervention of the past decade. But most of the conversation focuses on weight loss.
How Insulin Controls Blood Sugar
You eat two slices of white bread with lunch. You didn't add sugar to anything. But in the next 45 minutes, your blood sugar will spike higher.

Water Fasting — What Happens to Your Body
Most people have fasted for 16 hours and called it intermittent fasting. Water fasting is a different thing entirely. Here's what happens.

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
FAQ's
Sources
Spiegel K et al. — 'Impact of sleep debt on metabolic and endocrine function' (Lancet, 1999) — https://pubmed.ncbi.nlm.nih.gov/10543671/
Spiegel K et al. — 'Sleep curtailment in healthy young men is associated with decreased leptin levels' (PLOS Medicine, 2004) — https://pubmed.ncbi.nlm.nih.gov/15602591/
Donga E et al. — 'A single night of partial sleep deprivation induces insulin resistance' (JCEM, 2010) — https://pubmed.ncbi.nlm.nih.gov/20371664/
Van Cauter E et al. — 'Roles of circadian rhythmicity and sleep in human hormonal regulation' (Endocrine Reviews, 1997) — https://pubmed.ncbi.nlm.nih.gov/9159288/
Taheri S et al. — 'Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index' (PLOS Medicine, 2004) — https://pubmed.ncbi.nlm.nih.gov/15602591/

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
