9 April, 2026
Edition: Insulin & Blood Sugar
Vol 2, Edition 2
How Insulin Controls Blood Sugar — And Why Most People Have It Backwards
You eat two slices of white bread with lunch. You didn't add sugar to anything. But in the next 45 minutes, your blood sugar will spike higher than if you had eaten two teaspoons of table sugar. Here's exactly what happens — minute by minute — and why understanding it changes everything about how you eat.

Published By: MAP30 Challenge | Authored By: John Shaw
Article Directory: https://map30challenge.com/article-directory
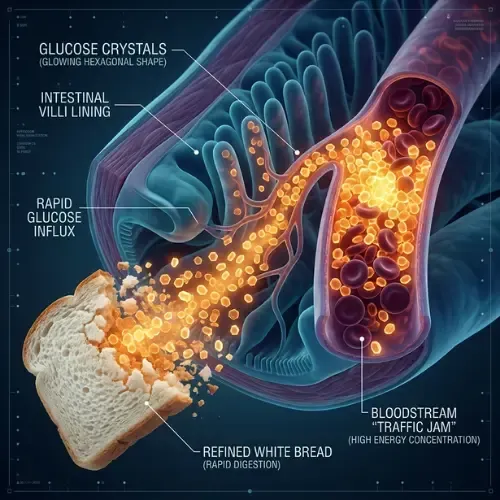
Two slices of white bread. No butter, no jam, no spread. Just bread.
White bread has a glycemic index of 70 to 75. Table sugar — pure sucrose — has a glycemic index of 65. That means two plain slices of white bread will raise your blood sugar faster and higher than the equivalent calories from sugar straight off the spoon. Not because bread contains more sugar than sugar. Because the refining process has already stripped away everything that would slow the glucose down — the fiber, the protein structure, the physical matrix of the grain — leaving a product that your digestive system converts to glucose almost instantaneously.
Most people don't know this. Most people think of bread as a neutral food and sugar as the enemy. The reality is more nuanced and more consequential: it is the speed of glucose delivery that matters, not just the source. And understanding that speed — and what insulin does in response to it — is the foundation of understanding why your body stores fat, why you're hungry two hours after a full meal, and why the standard dietary advice of the past fifty years has failed so comprehensively.
What Happens to Your Blood Sugar in 90 Minutes
Here is how I want you to think about this. At any given moment, your entire bloodstream contains about one teaspoon of glucose. That's it. One teaspoon. Not a cup, not a glass — a teaspoon. And your pancreas is working constantly, like a thermostat regulating the temperature in your home, to keep it right at that level. When glucose goes up — because you ate — your pancreas releases insulin to bring it back down to that one-teaspoon set point. When glucose goes down — because you haven't eaten — it releases glucagon to bring it back up. Every meal you eat is your pancreas scrambling to maintain that one teaspoon. Understanding that makes everything that follows make sense.
When you eat carbohydrates — any carbohydrates, from white bread to brown rice to fruit — the digestive process breaks them down into glucose. That glucose enters the bloodstream through the walls of the small intestine. How fast it enters depends almost entirely on what else was in the food: fiber slows it down, fat slows it down, protein slows it down. Refined carbohydrates stripped of all three deliver glucose rapidly. Whole foods deliver it slowly.
The moment glucose enters the bloodstream, blood sugar begins to rise. The pancreas detects this rise through glucose-sensing beta cells and begins releasing insulin in proportion to the rate and magnitude of the glucose increase. The faster and higher blood sugar rises, the larger and more rapid the insulin response.
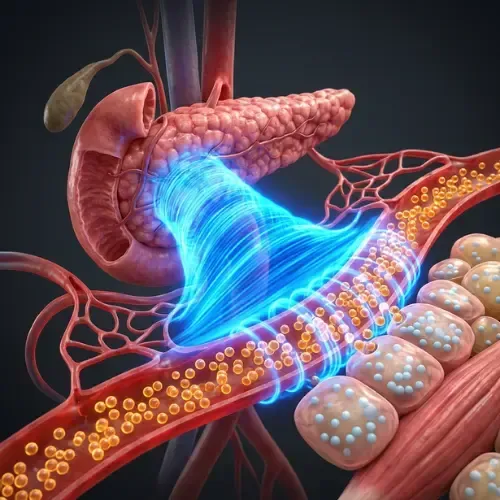
Here is the sequence that follows a high-glycemic meal — the kind most people eat several times a day:
Minutes 0–15: Glucose from the meal begins entering the bloodstream. Blood sugar starts rising. The pancreas releases a first phase of insulin — a rapid burst designed to prepare cells for incoming glucose.
Minutes 15–30: Blood sugar peaks. For a high-glycemic meal this peak can be substantial — 140 to 180 mg/dL or higher in people with any degree of insulin resistance. The pancreas is now releasing a second, sustained phase of insulin to clear the glucose from the bloodstream.
Minutes 30–45: Insulin is working. Glucose is being moved from the bloodstream into cells — primarily liver and skeletal muscle for glycogen storage, and fat cells for storage as triglycerides. Blood sugar begins falling.
Minutes 45–60: Blood sugar continues falling as insulin drives glucose clearance. In some people — particularly those with insulin resistance who required a large insulin response — blood sugar overshoots the baseline. This is reactive hypoglycemia: blood sugar drops below the fasting level, sometimes significantly.
Minutes 60–90: The glucose crash. Energy drops. Brain fog sets in. Concentration becomes difficult. And — most predictably — hunger returns. Not because the body needs fuel. Because blood sugar has dropped and the hormonal signals that follow a glucose crash are driving you back toward food.
This cycle — spike, insulin surge, crash, hunger — is not a character flaw or a lack of willpower. It is a predictable biochemical sequence driven by the composition of the food. Change the food and the sequence changes. Remove the spike and the crash never happens. Remove the crash and the hunger doesn't follow.
Research published in Pediatrics found that people who consumed high-glycemic meals showed significantly greater hunger and food intake at the next meal compared to those who consumed low-glycemic meals of equivalent calories — demonstrating that the glycemic response, not the caloric content, drives subsequent appetite. (Ludwig DS et al. — Pediatrics, 1999)
What Insulin Actually Is — And What It Actually Does
Insulin is a peptide hormone produced by beta cells in the pancreatic islets of Langerhans. It is released in response to rising blood glucose and its primary job is to facilitate the movement of glucose from the bloodstream into cells. Without insulin, glucose cannot enter most cells — it remains in the bloodstream, causing the damage associated with uncontrolled diabetes.
But insulin does considerably more than manage blood glucose. It is the body's primary anabolic hormone — the master signal for growth, storage, and energy conservation. When insulin is elevated, the body is in storage mode across multiple systems simultaneously:
In fat cells — insulin activates lipoprotein lipase, promoting fat uptake and storage, while suppressing hormone-sensitive lipase, preventing fat from being released for fuel. Elevated insulin locks fat in adipose tissue.
In the liver — insulin promotes glycogen synthesis and, when glycogen stores are full, de novo lipogenesis: the conversion of excess glucose into fat. The liver becomes a fat factory when insulin is chronically elevated.
In skeletal muscle — insulin drives glucose uptake for glycogen storage and energy use. Skeletal muscle is the largest insulin-sensitive tissue in the body and the primary site of glucose disposal in healthy metabolism.
In the brain — insulin receptors in the hypothalamus respond to insulin signals to regulate appetite and energy balance. Chronically elevated insulin contributes to the leptin resistance that impairs satiety signaling.
The critical point — the one most dietary advice ignores — is that insulin's fat storage effects are not incidental side effects of its blood sugar management function. They are its design. Insulin evolved in an environment where caloric surplus was rare and storing energy when it was available was a survival advantage. The problem is not insulin. The problem is chronic insulin elevation produced by a food environment that delivers glucose spikes multiple times a day, every day, for decades.
"Insulin is not the enemy. Chronic insulin elevation is. And chronic insulin elevation is not caused by insulin malfunctioning. It is caused by a diet that never lets it fall."
The Difference Between Blood Sugar and Insulin — Why One Hides the Other
This is the distinction that most people — and most physicians — miss. Blood sugar and insulin are not the same measurement. They do not move in parallel. And the one that is routinely tested — fasting glucose — is the last of the two to become abnormal.
Fasting glucose is a lagging indicator. When insulin resistance begins developing, the pancreas compensates by producing more insulin. For years, sometimes a decade or more, the extra insulin is sufficient to keep fasting glucose in the normal range. Blood sugar tests normal. The patient is told everything is fine. Meanwhile, fasting insulin is climbing, HOMA-IR is rising, fat is accumulating in the liver and visceral compartment, and the metabolic dysfunction is progressing silently.
Fasting insulin is the leading indicator. Fasting insulin reveals how hard the pancreas is working to maintain normal blood glucose. A fasting insulin of 15 μIU/mL alongside a fasting glucose of 90 mg/dL produces a HOMA-IR of 3.4 — firmly in insulin resistance territory — despite blood glucose that would be considered perfectly normal by any standard screening. The problem is invisible unless you look at both numbers together.
Postprandial blood sugar reveals what fasting glucose cannot. Even in people with normal fasting glucose, significant postprandial blood sugar spikes — the kind produced by high-glycemic meals — can occur for years before fasting glucose elevates. A continuous glucose monitor worn for two weeks will reveal the true glycemic response to every meal. Many people are genuinely shocked by what their blood sugar does after foods they considered healthy.
Only 12.2% of American Adults
have optimal metabolic health — defined as normal fasting glucose, blood pressure, triglycerides, HDL, and waist circumference without medica
Araújo J et al. — Metabolic Syndrome and Related Disorders, 2019
Why the Glycemic Index Matters — But Glycemic Load Matters More
The glycemic index ranks foods by how quickly they raise blood sugar relative to pure glucose. It is a useful concept but an incomplete one. A food's glycemic index tells you nothing about how much glucose it delivers — only how fast that glucose arrives.
Glycemic load corrects this by multiplying the glycemic index by the amount of carbohydrate in a serving. Watermelon has a high glycemic index — the sugars absorb quickly — but a low glycemic load because a serving contains relatively little carbohydrate. White bread has both a high glycemic index and a high glycemic load because it delivers a large amount of rapidly-absorbed glucose in a single serving.
For practical purposes, the most useful framework is simpler than either index: foods that have been industrially processed to remove fiber, protein structure, and the physical matrix that slows digestion will almost always produce a faster and larger blood sugar response than their whole-food equivalents. The processing is the variable. The fiber and food matrix are the brakes.

Some of the highest-glycemic foods are the ones most people consider harmless or even healthy:
White bread — glycemic index 70–75, higher than table sugar at 65
Rice cakes — glycemic index 82, one of the highest of any common food
Instant oatmeal — glycemic index 79, versus 55 for rolled oats — same grain, dramatically different response
Sports drinks — designed for rapid glucose delivery during athletic performance, not daily consumption
Most breakfast cereals — refined grain base with added sugar, glycemic index typically 70–85
Fruit juice — same sugar as whole fruit, none of the fiber that slows absorption
Low-fat flavored yogurt — fat removed and replaced with sugar to maintain palatability
The common thread: industrial processing has removed or destroyed the fiber and food matrix that would slow glucose absorption in the whole food version. The label says healthy. The glycemic response tells a different story.
What Chronic Blood Sugar Dysregulation Actually Looks Like
Most people experiencing blood sugar dysregulation don't experience it as a blood sugar problem. They experience it as fatigue, hunger, brain fog, and an inexplicable inability to control their eating. The connection between these symptoms and their metabolic cause is rarely made — because the symptoms are common enough to be normalized and the testing that would reveal the cause is rarely ordered.
The pattern is recognizable once you know what you're looking at. Energy that is entirely dependent on meal timing — present after eating, absent two hours later. Hunger that returns urgently within two to three hours of a full meal, driven not by actual energy deficit but by a glucose crash and the hormonal signals that follow it. Afternoon brain fog that lands reliably after a high-carbohydrate lunch. Difficulty concentrating in the late morning when blood sugar from breakfast has peaked and fallen. Cravings for sugar and refined carbohydrates that feel physiological — because they are.
"Blood sugar and insulin are not two separate topics. They are one mechanism — a lock and key system that determines whether your body is burning fuel or storing it. Understanding the sequence changes how you look at every meal you eat."
These are not symptoms of a broken metabolism. They are the predictable consequences of a metabolism running on an unstable fuel — one that spikes, triggers a hormone response, and crashes, over and over again, multiple times a day. And because the pancreas — that thermostat — is working every time to bring blood sugar back down to one teaspoon, insulin is never off duty. Glucagon never gets the call to come in and burn fat. The fat-burning window never opens. The solution is not more willpower or smaller portions. It is a fuel source that doesn't produce the spike in the first place.
This article is part of our Insulin & Blood Sugar series
Latest Articles

Diagnosed With Prediabetes. Now What?
Your fasting glucose came back at 99. Your doctor said everything looks fine. It isn't. Here's what that number actually means.

Non-Alcoholic Fatty Liver Disease
You don't drink. You're not overweight. You eat reasonably well. And your doctor just told you that your liver is showing signs of fat accumulation.

Water Fasting — What Happens to Your Body
Most people have fasted for 16 hours and called it intermittent fasting. Water fasting is a different thing entirely. Here's what happens.

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
FAQ's
1. How does insulin control blood sugar?
Insulin is released by the pancreas when blood glucose rises after eating. It binds to receptors on cells — primarily in the liver, skeletal muscle, and fat tissue — and signals them to take in glucose from the bloodstream. In fat cells, insulin promotes storage and simultaneously prevents stored fat from being released. When insulin falls between meals, the reverse becomes possible — fat can be released and burned for fuel. The pancreas manages this system continuously, keeping blood glucose at approximately one teaspoon at any given time.
2. What is the difference between blood sugar and insulin?
Blood sugar measures the concentration of glucose in the bloodstream at a given moment. Insulin measures the hormonal response to that glucose — how hard the pancreas is working to manage it. They do not become abnormal at the same time. Fasting insulin rises first as insulin resistance develops. Fasting glucose remains normal for years while insulin climbs, producing a false sense of metabolic health. Both numbers together — used to calculate HOMA-IR — give a far more accurate metabolic picture than either alone.
3. Why does white bread spike blood sugar more than sugar?
White bread has a glycemic index of 70 to 75, compared to 65 for table sugar. The refining process strips the fiber, protein, and food matrix that would slow digestion, leaving starch that breaks down and absorbs almost as rapidly as pure glucose. Two slices of white bread can produce a blood sugar response comparable to two teaspoons of table sugar — not because of its sugar content as labeled, but because of how fast the refined starch converts to glucose in the digestive tract.
4. What causes blood sugar spikes?
Foods that deliver glucose rapidly into the bloodstream — primarily refined carbohydrates and added sugars processed to remove the fiber and food matrix that would slow absorption. The magnitude of the spike depends on the glycemic load: how much rapidly-digestible carbohydrate was consumed and how quickly it absorbed. Protein, fat, and fiber all slow glucose absorption and reduce the spike. Eating refined carbohydrates without these buffers produces the sharpest responses.
5. What are the symptoms of blood sugar dysregulation?
Energy that rises and falls with meal timing; hunger returning urgently two to three hours after a full meal; afternoon brain fog after a high-carbohydrate lunch; difficulty concentrating in the late morning; strong cravings for sugar and refined carbohydrates; fatigue after meals; and poor sleep quality from overnight blood sugar fluctuations. These symptoms are common enough to be considered normal — but they are not normal. They are predictable consequences of a metabolism running on an unstable fuel source.
6. How do you stabilize blood sugar?
Remove refined carbohydrates and added sugars that produce sharp glucose spikes; prioritize protein and healthy fats at meals, which have low glycemic impact and slow absorption of any carbohydrates eaten alongside them; incorporate fiber-rich whole foods that blunt the glucose response; extend the overnight fasting window to allow insulin to fall fully between dinner and breakfast; and add brief movement after meals — even a 10-minute walk significantly reduces postprandial blood sugar spikes by driving glucose uptake in skeletal muscle.
Sources
Ludwig DS et al. — 'High glycemic index foods, overeating, and obesity' (Pediatrics, 1999) — https://pubmed.ncbi.nlm.nih.gov/10049134/
Araújo J et al. — 'Prevalence of Optimal Metabolic Health in American Adults' (Metabolic Syndrome and Related Disorders, 2019) — https://pubmed.ncbi.nlm.nih.gov/30484738/
Jenkins DJ et al. — 'Glycemic index of foods' (Am J Clin Nutr, 1981) — https://pubmed.ncbi.nlm.nih.gov/6259925/
Ebbeling CB et al. — 'Effects of a low glycemic load vs low fat diet in obese young adults' (JAMA, 2007) — https://pubmed.ncbi.nlm.nih.gov/17341711/
Tabák AG et al. — 'Prediabetes: a high-risk state for diabetes development' (Lancet, 2012) — https://pubmed.ncbi.nlm.nih.gov/22541686/

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
