Edition: Gut Health & Recovery
15 March, 2026
In The News
Vol 1, Edition 12
Why Chronic Inflammation Is Making You Fat
Chronic inflammation is not the redness and swelling of an injury. It is a silent, systemic biological state that disrupts hormones, impairs insulin signaling, and drives fat storage — often for years before a diagnosis is made. Here's how it works and what drives it.

Published By: MAP30 Challenge
Authored By: John Shaw
When most people hear the word inflammation, they think of an acute response — a sprained ankle swelling, a cut reddening as it heals. Acute inflammation is a healthy, functional biological process. It mobilizes immune cells to repair tissue and fight infection. It is temporary, localized, and self-limiting.
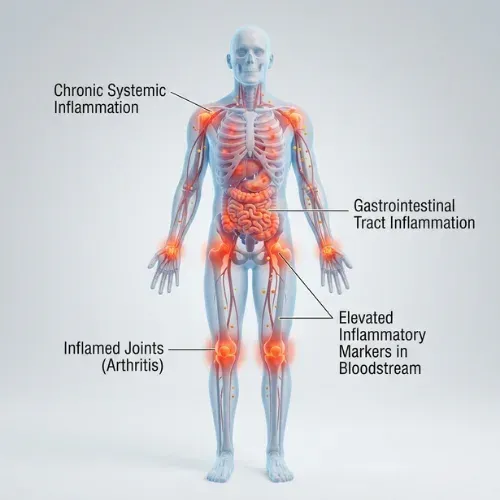
Chronic low-grade inflammation is something entirely different. It is not visible. It produces no swelling, no redness, no obvious pain. It operates at a systemic level — throughout the bloodstream, in adipose tissue, in the liver, in the walls of blood vessels — and it does so continuously, often for years or decades, without ever fully resolving.
What chronic inflammation does produce, consistently and measurably, is metabolic disruption. It impairs insulin signaling directly. It dysregulates the hormones that control hunger and fat storage. It accelerates visceral fat accumulation. It is an independent risk factor for type 2 diabetes, cardiovascular disease, metabolic syndrome, and obesity — and it sits upstream of all of them.
Acute vs. Chronic Inflammation: A Critical Distinction
Acute inflammation is the immune system doing its job. A pathogen enters, immune cells are dispatched, the area becomes inflamed, the threat is neutralized, the inflammation resolves. The entire process is designed to be temporary — activated by a trigger, resolved when the trigger is eliminated.
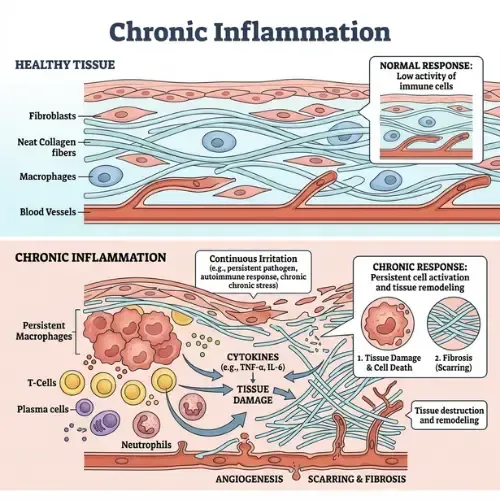
Chronic low-grade inflammation is what happens when the immune system's activation signal never fully turns off. Inflammatory cytokines — signaling proteins including TNF-alpha, IL-6, and C-reactive protein (CRP) — remain persistently elevated at levels too low to produce obvious symptoms but high enough to continuously interfere with normal cellular function.
The triggers for chronic inflammation are not pathogens. They are metabolic: excess visceral fat, chronically elevated blood glucose, gut barrier disruption, poor sleep, chronic psychological stress, ultra-processed food consumption, and sedentary behavior. These are conditions the immune system was not designed to resolve — because they are not infections. They are states. The inflammatory response stays activated because the trigger never goes away.
🔬 CLINICAL STUDY — Dandona P et al. (2004)
Trends in Immunology, 25(1), 4–7.
Research at the University of Buffalo established that obesity is associated with a state of chronic, low-grade systemic inflammation — characterized by elevated TNF-alpha, IL-6, and CRP — that is mechanistically distinct from acute inflammatory response. The authors demonstrated that this inflammatory state is not simply a consequence of excess fat, but an active contributor to the insulin resistance and metabolic dysfunction that drives further fat accumulation.
What This Means: Chronic inflammation and insulin resistance are bidirectional. Inflammation drives insulin resistance. Insulin resistance drives fat accumulation. Fat accumulation drives more inflammation. Breaking this cycle requires addressing the inflammatory state directly — not just the caloric balance.
How Chronic Inflammation Disrupts Hormones and Insulin
The connection between chronic inflammation and weight gain operates through several distinct hormonal pathways — each of which is independently documented and each of which reinforces the others.
Insulin resistance through cytokine interference
TNF-alpha — one of the primary pro-inflammatory cytokines elevated in chronic inflammation — directly interferes with insulin receptor signaling at the cellular level. It activates a kinase called IKK-beta which phosphorylates insulin receptor substrate proteins in a way that blocks the normal insulin signal cascade. In plain terms: TNF-alpha physically disrupts the molecular machinery through which insulin communicates with cells. This is one of the most well-established mechanisms linking inflammation to insulin resistance.
"Inflammation does not just make you sick. It makes your brain think you are starving while simultaneously preventing your body from accessing its stored energy."
Cortisol elevation and visceral fat
Chronic inflammation activates the HPA axis — the hormonal system governing the stress response — which elevates cortisol. As covered in the Cortisol article in this library, chronically elevated cortisol drives visceral fat accumulation through glucocorticoid receptor activation in abdominal adipose tissue, and simultaneously worsens insulin resistance. Visceral fat itself secretes inflammatory cytokines, creating a self-reinforcing loop: inflammation elevates cortisol, cortisol builds visceral fat, visceral fat generates more inflammation.
Leptin resistance
Inflammatory cytokines — particularly IL-6 and TNF-alpha — promote leptin resistance, a condition in which the brain stops responding normally to leptin's satiety signal. In a metabolically healthy person, leptin released by fat cells signals the hypothalamus that energy stores are sufficient and suppresses appetite. When inflammation disrupts this signal, the brain remains in a state of perceived energy deficit regardless of actual fat stores. Hunger persists. Eating continues. The excess energy is stored in an already inflamed, insulin-resistant metabolic environment.
🔬 CLINICAL STUDY — Hotamisligil GS et al. (1993)
Science, 259(5091), 87–91.
Groundbreaking research at Harvard Medical School demonstrated for the first time that TNF-alpha is expressed by adipose tissue and that its expression is elevated in obese animals and humans. Neutralizing TNF-alpha activity improved insulin sensitivity significantly. This paper established the mechanistic link between inflammation — specifically cytokine expression from fat tissue — and insulin resistance, fundamentally changing the scientific understanding of obesity and metabolic disease.
What This Means: Fat tissue is not metabolically inert — it is an active endocrine organ secreting inflammatory compounds that directly cause insulin resistance. The more visceral fat, the more inflammation, the worse the insulin resistance. This cycle, once established, is self-perpetuating.
The Gut Connection: Where Most Chronic Inflammation Starts

For a substantial portion of people with chronic low-grade inflammation, the origin is the gut — specifically, increased intestinal permeability driven by microbiome disruption.
As covered in the Gut Microbiome article in this library, a disrupted microbiome allows bacterial fragments — particularly lipopolysaccharides (LPS) from gram-negative bacteria — to cross the gut barrier and enter the bloodstream. The immune system responds to these bacterial fragments as a threat, initiating an inflammatory response. Because the gut barrier disruption is ongoing rather than transient, so is the inflammatory response. This is metabolic endotoxemia — a condition of chronic, low-level bacterial-driven inflammation that is now recognized as a primary driver of insulin resistance and metabolic disease.
Signs of Chronic Low-Grade Inflammation:
• Persistent fatigue that does not resolve with rest• Unexplained weight gain, particularly around the abdomen, that does not respond to diet and exercise• Joint stiffness and diffuse aching without clear injury• Brain fog — difficulty concentrating, slow mental processing• Frequent illness or slow recovery from illness• Skin conditions including eczema, psoriasis, or persistent acne• Elevated CRP, ESR, or fasting insulin on blood tests• Poor sleep quality despite adequate durationNote: These symptoms are non-specific and overlap with many conditions. Elevated inflammatory markers on blood tests provide the most reliable confirmation.
What Drives Chronic Inflammation — The Primary Sources
Chronic inflammation does not arise from a single cause. It is typically maintained by multiple simultaneous inputs — each contributing to the inflammatory burden and each potentially addressable.
1. Visceral fat
Visceral adipose tissue — the deep abdominal fat surrounding organs — is metabolically active and secretes pro-inflammatory cytokines including TNF-alpha, IL-6, and resistin continuously. More visceral fat means more systemic inflammation. Visceral fat reduction produces measurable decreases in inflammatory markers within weeks.
2. Ultra-processed food and refined sugar
Refined sugars drive chronic glycemic variability and insulin elevation. Emulsifiers in ultra-processed foods disrupt the gut barrier, as documented in the Dirty Secret of Ultra-Processed Foods article in this library. Processed seed oils — high in omega-6 fatty acids — provide substrates for pro-inflammatory eicosanoid production. The combination creates a dietary pattern that sustains inflammation through multiple pathways simultaneously.
3. Poor sleep
Sleep deprivation elevates inflammatory markers including CRP and IL-6 measurably after even a single night of poor sleep. Chronic sleep restriction is an independent driver of elevated TNF-alpha and is associated with significantly higher inflammatory burden in population studies. The mechanism involves both HPA axis dysregulation and impaired nighttime immune regulation.
4. Chronic psychological stress
Psychological stress activates the sympathetic nervous system and HPA axis, producing sustained cortisol and catecholamine elevation. These hormones have direct pro-inflammatory effects over time, and chronic stress is independently associated with elevated CRP and IL-6 in otherwise healthy adults.
5. Sedentary behavior
Regular physical activity has well-documented anti-inflammatory effects through multiple pathways — including IL-6 release from contracting muscle that paradoxically acts as an anti-inflammatory signal, and improved insulin sensitivity that reduces the inflammatory burden from glycemic dysregulation. Sedentary behavior removes these protective mechanisms.
Mitochondrial dysfunction — covered in depth in the How Mitochondria Work article in this library — is both a cause and consequence of chronic inflammation. Damaged mitochondria release signals that activate the same inflammatory pathways driven by visceral fat and gut disruption, creating another self-reinforcing loop that exercise and dietary change both address.
How to Reduce Chronic Inflammation — What the Evidence Supports
Because chronic inflammation is maintained by multiple simultaneous inputs, the most effective approach addresses several of them concurrently rather than targeting a single variable. The interventions with the strongest and most consistent evidence:

Remove ultra-processed foods and refined sugar
This addresses three inflammatory inputs simultaneously: gut barrier disruption from emulsifiers, glycemic variability from refined carbohydrates, and omega-6 excess from processed seed oils. Research consistently shows measurable CRP reductions within two to four weeks of dietary pattern change in this direction.
Prioritize sleep quality and duration
Consistent seven to nine hours of quality sleep normalizes the cortisol diurnal rhythm, reduces sympathetic nervous system activation, and allows the nighttime immune regulation processes that suppress chronic inflammatory signaling to function properly. Sleep improvement is one of the fastest interventions for reducing inflammatory markers in clinical settings.
Reduce visceral fat
Visceral fat reduction — achieved most efficiently through carbohydrate restriction and fasting protocols that lower insulin — directly reduces the primary source of TNF-alpha and IL-6 in most metabolically compromised adults. As visceral fat decreases, inflammatory burden decreases. The relationship is dose-dependent and bidirectional.
Incorporate resistance and aerobic exercise
Exercise-induced IL-6 from contracting skeletal muscle has anti-inflammatory effects that differ from the chronic IL-6 elevation seen in metabolic disease. Regular moderate exercise reduces baseline CRP, improves insulin sensitivity, and reduces visceral fat — addressing inflammation through three separate mechanisms simultaneously.
What This Means for the MAP30 Challenge
Chronic inflammation is not a separate condition from insulin resistance, metabolic syndrome, or obesity — it is deeply intertwined with all of them, often sitting at the root of the cascade that produces them. The MAP30 framework addresses the metabolic environment that sustains inflammation: chronic insulin elevation, poor sleep, ultra-processed food consumption, gut disruption, and visceral fat accumulation.
For many people who have struggled with weight loss despite doing everything they were told to do, chronic inflammation is the variable that has not been addressed. It is not visible on a scale. It does not show up in a basic blood panel. But it is measurable — through fasting insulin, CRP, and waist circumference — and it responds to the same metabolic interventions that MAP30 is built around.
| What the Label Said | What It Often Meant |
|---|---|
| Fat-free | Sugar and refined carbohydrates substituted for fat to maintain palatability. |
| Low fat | Calorie content often similar; sugar content frequently higher than the full-fat version. |
| Reduced fat | Fat replaced by added sugars, starches, or carbohydrate-based thickeners. |
| Light | Typically reduced fat or calories, often with a corresponding increase in sugar or sodium. |
| Principle | What the Current Evidence Supports |
|---|---|
| Stop avoiding all fat | Unsaturated fats from whole food sources are associated with better outcomes. The undifferentiated avoidance of fat is not supported by current evidence. |
| Reduce refined carbohydrates and added sugar | White bread, processed grain products, and added sugar drive insulin response and liver fat accumulation. The pyramid's grain endorsement did not make this distinction — and it matters. |
| Prioritize whole foods over processed | Much of the harm in the low-fat era came from processed products where fat was replaced with sugar. Ultra-processed foods, regardless of fat content, are independently associated with worse health outcomes. |
| Emphasize protein and fiber | Both slow digestion, support satiety, blunt insulin response, and help preserve muscle mass — all underemphasized in the pyramid era. |
| Read ingredient labels, not just nutrition labels | If sugar or refined flour appears early in a product's ingredient list, its health marketing deserves scrutiny regardless of fat content. |
| Treat guidelines as starting points | The 2025–2030 guidelines represent a substantially different view than 1992. Nutritional guidance evolves as evidence accumulates — staying informed matters more than following any single era's advice. |
This article is part of our Gut Health & Recovery series.
Link Here: Gut Health & Recovery
Latest Articles
How to Lower Cortisol
You’ve been told stress makes you eat more. That’s only part of the story. Cortisol directly instructs your body to store fat...
Metabolic Syndrome
Most people who have metabolic syndrome don’t know it. There’s no single dramatic symptom — just a cluster of five...

Ozempic & GLP-1
GLP-1 drugs work. That part is true. What the headlines skip is how they work, what happens when you stop...

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
Disclaimer: The information in this article is for educational and historical context only and does not constitute medical or dietary advice. Nutritional science is an evolving field and individual dietary needs vary. Consult a qualified healthcare provider or registered dietitian before making significant changes to your diet or health management plan.
FAQ's
1. What is chronic low-grade inflammation?
Chronic low-grade inflammation is a persistent, systemic state of immune activation characterized by continuously elevated inflammatory markers — TNF-alpha, IL-6, and CRP — at levels too low to produce obvious symptoms but sufficient to disrupt insulin signaling, dysregulate hunger hormones, and accelerate metabolic disease. Unlike acute inflammation from injury or infection, it does not resolve because its triggers — visceral fat, poor diet, gut disruption, poor sleep — are ongoing states, not temporary events.
2. How does chronic inflammation cause weight gain?
Through three primary mechanisms: TNF-alpha directly blocks insulin receptor signaling, contributing to insulin resistance and fat storage; inflammatory activation of the HPA axis elevates cortisol, which drives visceral fat accumulation; and inflammatory cytokines promote leptin resistance, impairing the brain's satiety signal so hunger persists despite adequate energy stores. Each mechanism independently promotes fat accumulation, and they reinforce each other.
3. What is the connection between chronic inflammation and hormones?
Chronic inflammation dysregulates multiple metabolic hormones simultaneously. It promotes insulin resistance through TNF-alpha interference with insulin receptor signaling. It elevates cortisol through HPA axis activation. It impairs leptin signaling, suppressing satiety. It reduces adiponectin — an anti-inflammatory, insulin-sensitizing hormone secreted by fat cells. The hormonal environment created by chronic inflammation is one that consistently promotes fat storage and resists fat mobilization.
4. What are the signs of chronic inflammation in the body?
Common signs include persistent fatigue unresolved by rest, unexplained abdominal weight gain unresponsive to diet and exercise, brain fog and difficulty concentrating, frequent illness or poor recovery, joint stiffness without injury, and poor sleep quality despite adequate duration. Blood markers — elevated CRP, ESR, and fasting insulin — provide more reliable confirmation. Many of these signs overlap with insulin resistance because the two conditions are so frequently co-present and mutually reinforcing.
5. What foods cause chronic inflammation?
The dietary patterns most consistently associated with elevated chronic inflammatory markers are: high refined carbohydrate and added sugar intake, which drives glycemic variability and insulin elevation; ultra-processed foods, whose emulsifiers disrupt the gut barrier and whose refined oils provide omega-6 substrates for pro-inflammatory compounds; and low dietary fiber, which starves the butyrate-producing gut bacteria that maintain intestinal barrier integrity and suppress inflammatory signaling.
6. How do you reduce chronic inflammation naturally?
The most evidence-supported interventions are: eliminating ultra-processed foods and refined sugar to reduce the primary dietary inflammatory inputs; prioritizing consistent quality sleep of seven to nine hours; reducing visceral fat through carbohydrate restriction and fasting protocols; incorporating regular moderate exercise; and supporting gut barrier integrity through dietary fiber diversity and fermented foods. These approaches address inflammation through multiple simultaneous pathways — which is necessary because chronic inflammation is maintained by multiple simultaneous inputs.
Sources
Hotamisligil GS et al. — 'Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance' (Science, 1993)
https://pubmed.ncbi.nlm.nih.gov/7678183/
Dandona P et al. — 'Inflammation: the link between insulin resistance, obesity and diabetes' (Trends in Immunology, 2004)
https://pubmed.ncbi.nlm.nih.gov/14698282/
Calder PC et al. — 'Dietary factors and low-grade inflammation in relation to overweight and obesity' (British Journal of Nutrition, 2011)
https://pubmed.ncbi.nlm.nih.gov/22062335/
Cani PD et al. — 'Metabolic Endotoxemia Initiates Obesity and Insulin Resistance' (Diabetes, 2007)
https://pubmed.ncbi.nlm.nih.gov/17519319/
Ridker PM — 'C-reactive protein and the prediction of cardiovascular events' (New England Journal of Medicine, 2007)
https://pubmed.ncbi.nlm.nih.gov/17267907/
Pedersen BK, Febbraio MA — 'Muscle as an endocrine organ: focus on muscle-derived interleukin-6' (Physiological Reviews, 2008)

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
