3 April, 2026
Edition: Hormones & Metabolism
Vol 1, Edition 20
Glucagon Benefits for Fat Loss: The Hormone That Unlocks Stored Energy
Every conversation about blood sugar and fat loss focuses on insulin. But insulin has a counterpart that does the opposite job — and understanding it may be more important for fat loss than anything you've been told about calories.
Published By: MAP30 Challenge
Authored By: John Shaw
Insulin vs. Glucagon: The Hormonal Seesaw
Insulin vs. Glucagon: The Hormonal Seesaw
Fat Storage Mode — Insulin Dominant
What's happening in your body right now if you've eaten recently
Glucagon suppressed
Insulin directly inhibits alpha cells in the islets of Langerhans, preventing glucagon release.
Fat cells locked
Insulin suppresses hormone-sensitive lipase (HSL). Stored fatty acids cannot leave adipose tissue.
Fuel source: glucose
Body burns incoming food. Liver stores excess glucose as glycogen. Fat stores untouched.
Liver in storage mode
Glycogen synthesis active. Ketone production off. Fatty acid oxidation minimal.
Growth hormone suppressed
Fat mobilization and lean tissue repair signals downregulated while insulin is elevated.
Fat Burning Mode — Glucagon Rising
What's happening after 12–16 hours of fasting or sustained carbohydrate restriction
Glucagon activated
Falling insulin removes inhibition from alpha cells. Glucagon rises and sends fat-release signals throughout the body.
Fat cells unlocked
Glucagon activates HSL. Stored fatty acids stream out of adipose tissue into the bloodstream.
Fuel source: fat + ketones
Liver converts fatty acids to ketones. Brain and muscles switch to running on stored energy.
Liver in fat-burning mode
Glycogenolysis then ketogenesis active. Glucagon upregulates fatty acid oxidation genes in liver cells.
Growth hormone rising
Supports fat mobilization, lean tissue preservation, and metabolic repair during the fasted state.
Insulin gets the attention. Every conversation about metabolic health, fat storage, and blood sugar regulation eventually lands on insulin — how it works, what happens when it doesn't, why it matters. And all of that is correct. Insulin is central.
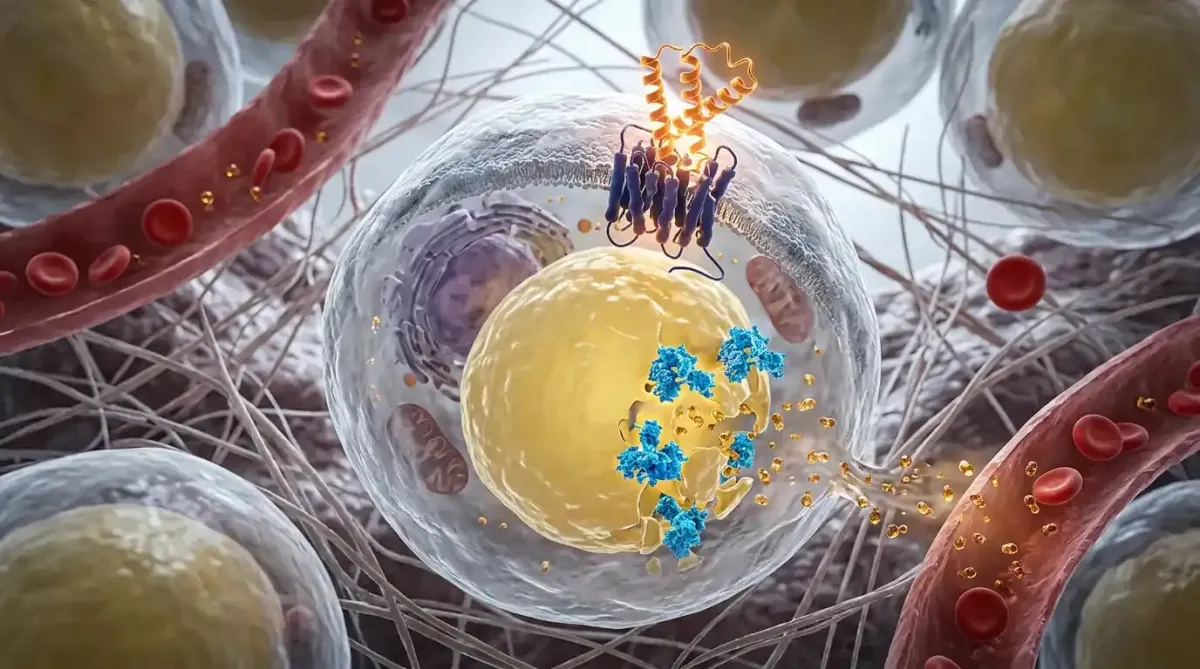
But insulin does not work alone. It works in opposition to another pancreatic hormone that most people outside of endocrinology have barely heard of: glucagon. Where insulin drives energy storage, glucagon drives energy release. Where insulin tells the liver to store glucose, glucagon tells it to release stored glucose and produce ketones from fat. Where insulin locks fat in adipose tissue, glucagon unlocks it.
The seesaw between these two hormones is the mechanism behind fat burning. Not calories. Not willpower. The ratio of glucagon to insulin at any given moment determines whether your body is accessing stored energy or adding to it. Understanding how glucagon works — and what suppresses and activates it — is one of the most useful and underutilized pieces of metabolic knowledge available to anyone trying to change their body composition.
"Insulin and glucagon are not just hormones. They are the on/off switch for your body's stored energy. When insulin is high, the switch is off. When glucagon rises and insulin falls, the switch is on."
What Is Glucagon and What Does It Do
Glucagon is a peptide hormone produced by alpha cells in the islets of Langerhans in the pancreas — the same tissue cluster that produces insulin from beta cells. Insulin and glucagon are released by neighboring cells, and their secretion is regulated in opposition: conditions that stimulate insulin release tend to suppress glucagon, and conditions that suppress insulin tend to allow or stimulate glucagon.
Its primary jobs are three:
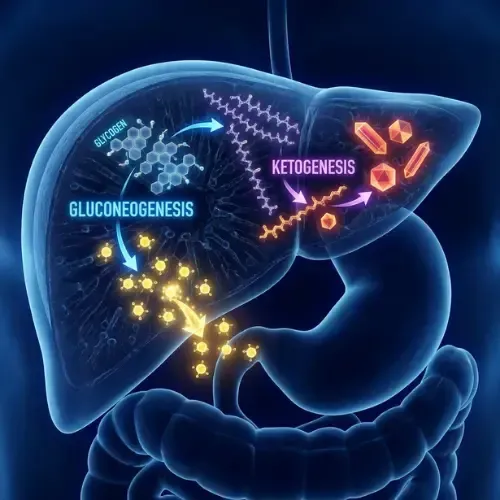
1. Glycogenolysis — Glucagon signals the liver to break down stored glycogen — the liver's glucose reserve — and release glucose into the bloodstream. This is the hormone's fastest response. When blood glucose falls between meals or during fasting, glucagon maintains blood sugar within a functional range by mobilizing the liver's glycogen stores. This is what prevents hypoglycemia during an overnight fast.
2. Gluconeogenesis — When glycogen is depleted — typically after 12 to 16 hours of fasting — glucagon shifts the liver toward gluconeogenesis: the production of new glucose from non-glucose substrates, primarily amino acids and glycerol. This is a secondary mechanism that sustains blood glucose when glycogen is exhausted and food is unavailable.
3. Lipolysis and ketogenesis — This is glucagon's most important function for fat loss. Glucagon activates hormone-sensitive lipase (HSL) in adipose tissue — the same enzyme that insulin suppresses. When glucagon rises and insulin falls, HSL is freed to release stored fatty acids from fat cells. The liver then converts these fatty acids into ketones, providing an alternative fuel for the brain and body. This is the mechanism of fat burning in fasted states.
"Glucagon is not merely a counter-regulatory hormone. It is an active participant in the metabolic switch from glucose oxidation to fat oxidation." — Müller TD et al. — Molecular Metabolism, 2017
Glucagon and Insulin — Why the Ratio Matters More Than Either One Alone
The relationship between glucagon and insulin is not additive — it is antagonistic. They do not cooperate. They compete. The metabolic state of the body at any given time is determined not by absolute glucagon or insulin levels but by the ratio between them.
When insulin is high — after a carbohydrate-rich meal, during chronic insulin resistance, with frequent eating patterns — glucagon's signaling is suppressed even if glucagon itself is present. Insulin directly inhibits glucagon release from alpha cells through paracrine signaling within the islets of Langerhans. And even when glucagon is released, its downstream signaling on fat cells is blocked while insulin is active — because insulin's signal to suppress HSL overrides glucagon's signal to activate it.
This is why the fed state is categorically different from the fasted state — not as a matter of degree but of kind. In the fed state with elevated insulin, glucagon's fat-releasing signal is essentially silenced. In the fasted state with low insulin, glucagon's signal can operate. The fat burning is not enhanced — it is enabled.

The three states this ratio produces in practice:
Fed state (insulin high): Glucagon suppressed. Hormone-sensitive lipase inhibited. Fat cells locked. Liver storing glycogen. Body burning incoming glucose from the meal.
Early fasting (insulin falling): Glucagon beginning to rise. Hormone-sensitive lipase activating. Liver glycogen being mobilized. Blood sugar maintained without incoming food.
Extended fasting (insulin low): Glucagon elevated. Full lipolysis activated. Fatty acids streaming from fat cells. Liver producing ketones. Brain and muscles running on stored fat.
The transition between these states takes 10–16 hours of consistent fasting and is the metabolic switch at the center of everything MAP30 teaches.
Glucagon Benefits for Fat Loss — What the Research Shows
The fat loss benefits of optimizing the glucagon-to-insulin ratio are not theoretical. They are the mechanism behind every dietary and fasting intervention that produces durable fat loss. Three findings are particularly relevant.
A 2019 review in Cell Metabolism by Müller and colleagues established that glucagon's role in lipid metabolism is substantially more active than previously understood. Beyond simply allowing HSL to function, glucagon directly upregulates genes involved in fatty acid oxidation in liver cells — accelerating the rate at which fatty acids are converted to ketones once they arrive. This means glucagon does not merely permit fat burning; it actively accelerates the downstream processing of mobilized fat.
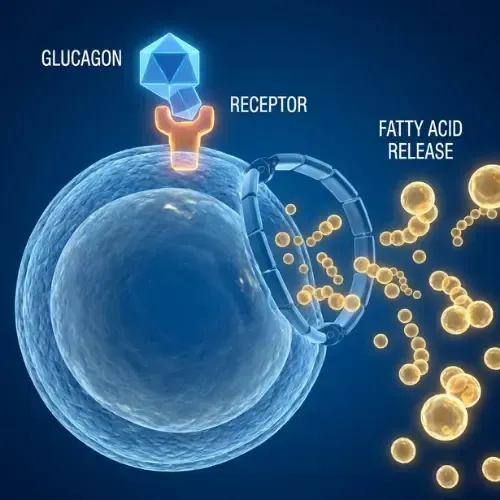
Research on protein-induced glucagon secretion — documented in multiple metabolic ward studies — demonstrates that dietary protein stimulates significant glucagon release alongside its insulin response. The insulin-to-glucagon ratio after a high-protein meal is substantially lower than after a high-carbohydrate meal of equivalent calories. This is part of the mechanism behind protein's superior satiety and fat loss effects compared to carbohydrates at equivalent caloric content: it stimulates glucagon alongside insulin rather than insulin alone.
Studies on intermittent fasting and time-restricted eating consistently show that the metabolic improvements — reduced visceral fat, improved insulin sensitivity, reduced triglycerides, increased fat oxidation — are mediated substantially through the regular activation of glucagon's fat-mobilizing pathway. The fasting window is not primarily a calorie restriction mechanism. It is a glucagon activation mechanism.
"Every hour of fasting is an hour of rising glucagon-to-insulin ratio. Every hour of that ratio rising is an hour of fat burning biology becoming progressively more active."
Glucagon and the Natural GLP-1 Connection
There is a direct relationship between glucagon signaling and GLP-1 — the gut hormone discussed in depth in the Ozempic and GLP-1 article in this library. GLP-1 suppresses glucagon as part of its mechanism: after eating, GLP-1 rises, stimulates insulin, and simultaneously suppresses glucagon to prevent inappropriate glucose production while a meal is being absorbed. This is appropriate and functional in healthy metabolism.
The problem arises in insulin-resistant metabolism where GLP-1 signaling is chronically impaired. When GLP-1 response is blunted, glucagon suppression after meals is incomplete — contributing to the inappropriately elevated glucagon levels seen in type 2 diabetes, which worsen postprandial hyperglycemia. This is one of the reasons GLP-1 drugs like Ozempic have a glucagon-suppressing effect as part of their mechanism.
The natural approach — restoring GLP-1 sensitivity through dietary change, gut microbiome support, and fasting protocols — also normalizes the glucagon-GLP-1 relationship, producing appropriate glucagon suppression after meals and appropriate glucagon activation during fasting periods. The goal is not to maximize glucagon. It is to restore the dynamic oscillation between glucagon and insulin that a functional metabolic environment produces automatically.
This article is part of our Hormones & Metabolism series. Hormones & Metabolism
Latest Articles

24-Hour Fasting Benefits
A 16-hour fast and a 24-hour fast are not just different in duration — they produce different metabolic events. Here's what changes in that second half of the day.
Visceral Fat vs. Subcutaneous Fat
Not all body fat is equal — and the difference matters more than almost anything else in metabolic health. One type stores energy quietly.

What Is HOMA-IR
Most people with insulin resistance have normal blood sugar. That's exactly why the standard blood sugar test misses it. HOMA-IR — a calculation using fasting insulin and fasting glucose

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
FAQ's
1. What is glucagon and what does it do?
Glucagon is a hormone produced by alpha cells in the pancreas that does the opposite job of insulin. While insulin drives energy storage, glucagon drives energy release. Its three primary functions are: glycogenolysis — signaling the liver to release stored glucose; gluconeogenesis — producing new glucose from amino acids and glycerol when glycogen is depleted; and lipolysis — activating hormone-sensitive lipase to release stored fatty acids from fat cells, which the liver converts to ketones for fuel.
2. What are the benefits of glucagon for fat loss?
Glucagon is the primary hormonal signal that enables fat burning. When glucagon rises and insulin falls — during fasting or carbohydrate restriction — hormone-sensitive lipase is freed to release stored fatty acids from fat cells. Glucagon also upregulates genes involved in fatty acid oxidation in liver cells, actively accelerating the processing of mobilized fat. Without glucagon activation, the fat burning pathway remains biochemically suppressed regardless of caloric intake.
3. What is the relationship between insulin and glucagon?
Insulin and glucagon are directly antagonistic — they suppress each other and produce opposite metabolic effects. When insulin is elevated, glucagon secretion is inhibited and its downstream fat-releasing signal is blocked. When insulin falls during fasting or carbohydrate restriction, glucagon can rise and its fat-mobilizing effects can operate. The glucagon-to-insulin ratio — not the absolute level of either hormone — determines whether the body is in fat storage or fat burning mode at any given time.
4. How does fasting increase glucagon?
During fasting, blood glucose falls gradually as glycogen is consumed. The falling glucose signal directly stimulates alpha cells in the pancreas to release glucagon. Simultaneously, insulin falls as glucose intake stops — removing the paracrine inhibition that insulin exerts on alpha cells within the islets of Langerhans. Both mechanisms converge to allow glucagon to rise. This is why the fat-burning effects of fasting accelerate progressively — glucagon increases as the fast extends.
5. Does protein affect glucagon?
Yes — dietary protein is a significant direct stimulator of glucagon secretion, independent of its insulin-stimulating effect. Amino acids from protein stimulate alpha cell glucagon release alongside beta cell insulin release. The result is that after a high-protein meal, the insulin-to-glucagon ratio is substantially lower than after a high-carbohydrate meal of equivalent calories. This is part of the mechanism behind protein's superior satiety and fat loss effects — it supports glucagon signaling alongside insulin, rather than suppressing it.
6. Can you have too much glucagon?
Yes — chronically elevated glucagon, as seen in poorly controlled type 2 diabetes, contributes to excessive hepatic glucose production and worsening hyperglycemia. In these patients, the normal GLP-1-mediated suppression of glucagon after meals is impaired, allowing glucagon to remain inappropriately elevated after eating. The goal is not to maximize glucagon but to restore the dynamic oscillation — appropriately suppressed after meals, appropriately active during fasting — that characterizes healthy metabolic function.
Sources
Müller TD et al. — 'Glucagon-like peptide 1 (GLP-1)' (Molecular Metabolism, 2019) — https://pubmed.ncbi.nlm.nih.gov/31068332/
Holst JJ et al. — 'The incretin system and its role in type 2 diabetes mellitus' (Molecular & Cellular Endocrinology, 2009) — https://pubmed.ncbi.nlm.nih.gov/19289146/
Cahill GF — 'Fuel metabolism in starvation' (Annual Review of Nutrition, 2006) — https://pubmed.ncbi.nlm.nih.gov/16848698/
Unger RH, Cherrington AD — 'Glucagonocentric restructuring of diabetes' (Journal of Clinical Investigation, 2012) — https://pubmed.ncbi.nlm.nih.gov/22232217/
Anton SD et al. — 'Flipping the Metabolic Switch' (Obesity, 2018) — https://pubmed.ncbi.nlm.nih.gov/29086496/

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
