Edition: Gut Health & Recovery
30 March, 2026
In The News
Vol 1, Edition 23
How to Improve Your Gut Microbiome Naturally — And Why It Fixes More Than Your Digestion
You have more bacterial cells in your gut than human cells in your entire body. Most people know the gut microbiome exists. Almost nobody understands what it actually does — or how profoundly its disruption drives insulin resistance, inflammation, and metabolic disease.
Published By: MAP30 Challenge
Authored By: John Shaw
When I was working through the early stages of reversing my own metabolic dysfunction, I kept hitting a wall. The dietary changes were in place. The fasting windows were consistent. The numbers were moving — but more slowly than the science suggested they should. What I eventually came to understand was that gut health was the variable I had not yet addressed. The microbiome was undermining the interventions from underneath, producing inflammation and impairing insulin signaling through pathways that had nothing to do with what I was eating at any given meal. Fixing the gut did not replace the other work. It made the other work actually work.
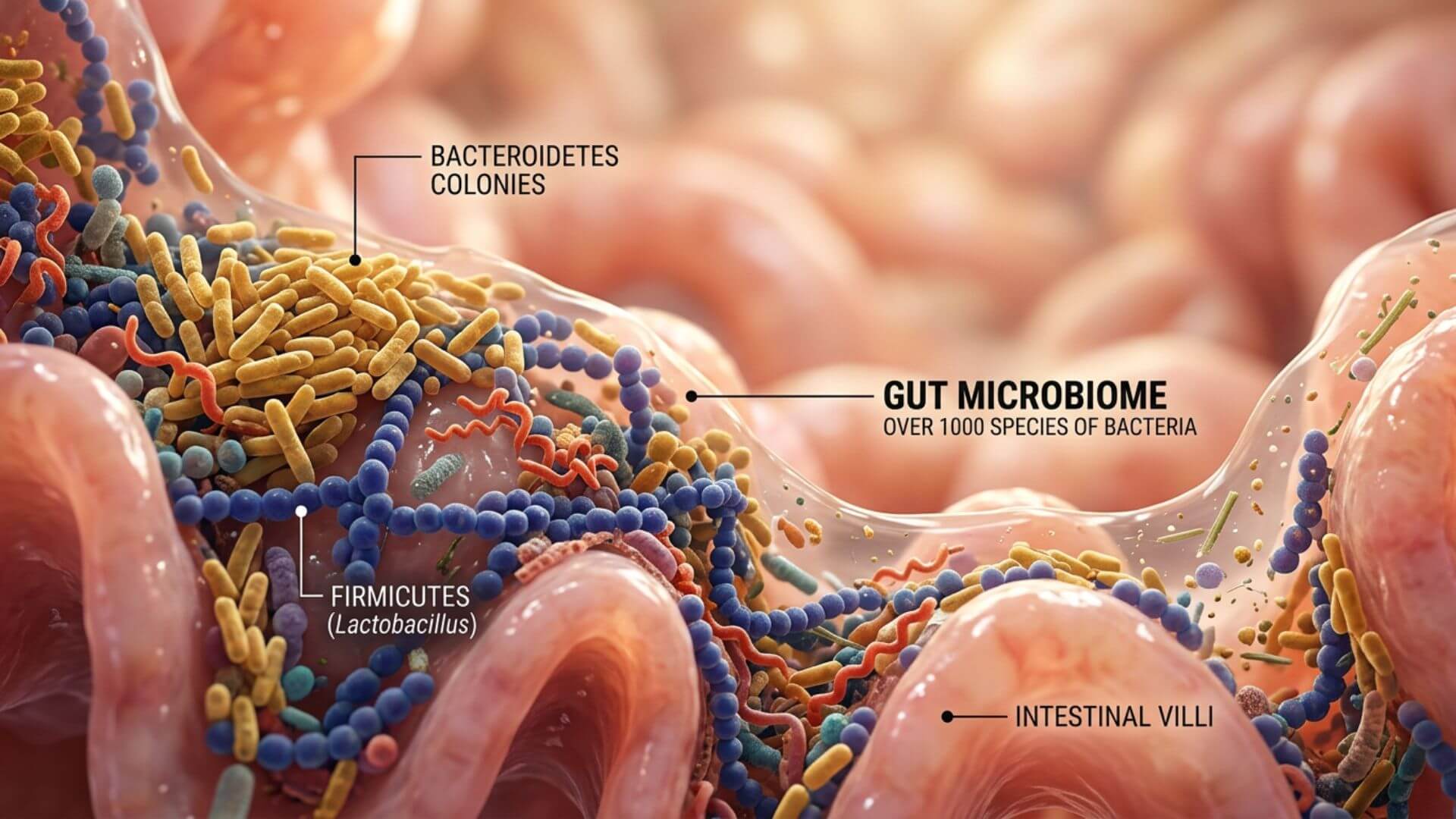
The gut is not just a digestive organ. It is one of the most metabolically active systems in the body — producing neurotransmitters, training the immune system, regulating inflammation, and directly influencing how cells respond to insulin. The average human gut contains approximately 38 trillion bacterial cells, over 1,000 distinct species, and a total microbial mass of roughly 1.5 kilograms — sometimes called the body's forgotten organ. Most people have heard that gut health matters. Far fewer understand the specific mechanisms by which a disrupted microbiome drives the same metabolic dysfunction that underlies insulin resistance, type 2 diabetes, obesity, and metabolic syndrome.
"The gut microbiome weighs as much as the liver, produces compounds that rival the most potent metabolic hormones, and communicates directly with the brain. It is not a secondary player in metabolic health. It is a primary one."
What the Gut Microbiome Actually Does
The gut microbiome is the community of trillions of microorganisms — bacteria, fungi, viruses, and other microbes — living primarily in the large intestine. Each person's microbiome is as unique as a fingerprint, shaped by birth method, early diet, antibiotic exposure, and decades of dietary choices. Its functions extend far beyond digestion:
Ferments dietary fiber into short-chain fatty acids (SCFAs) — particularly butyrate, propionate, and acetate — which serve as primary fuel for intestinal cells, regulate blood sugar, reduce inflammation, and influence fat storage
Trains and calibrates the immune system — approximately 70% of the immune system is located in gut-associated lymphoid tissue, and the microbiome is its primary educator
Produces neurotransmitters — including over 90% of the body's serotonin and significant quantities of dopamine precursors — through the gut-brain axis
Regulates intestinal barrier integrity — maintaining the tight junctions between gut cells that prevent bacterial toxins from entering the bloodstream
Modulates metabolic hormones — including GLP-1, PYY, and ghrelin — directly influencing appetite, satiety, and glucose metabolism
This is not a peripheral support system. The gut microbiome is a primary metabolic organ. Disrupting it disrupts all of these functions simultaneously — which is why gut dysbiosis produces consequences that look nothing like a digestive problem.
How a Disrupted Microbiome Drives Metabolic Disease — The Cascade
The connection between gut dysbiosis — a disrupted, low-diversity microbiome — and metabolic disease is not a single pathway. It is a cascade where one disruption triggers the next:
| Step | What Happens | Metabolic Consequence |
|---|---|---|
| 1 | Microbiome diversity decreases — fewer beneficial species, more pathogenic ones | SCFA production falls. Less butyrate means intestinal cells are underfed and the gut barrier weakens. |
| 2 | Gut barrier permeability increases ('leaky gut') | Bacterial fragments — particularly lipopolysaccharides (LPS) — cross into the bloodstream. |
| 3 | LPS triggers systemic immune response | Chronic low-grade inflammation develops. TNF-alpha and IL-6 levels rise. |
| 4 | Inflammatory cytokines impair insulin signaling | Insulin receptors in muscle and liver cells become less responsive — insulin resistance develops. |
| 5 | Pancreas compensates with more insulin | Hyperinsulinemia promotes fat storage, blocks fat burning, drives further metabolic dysfunction. |
| 6 | Metabolic syndrome criteria accumulate | Elevated triglycerides, visceral fat, blood pressure, fasting glucose — driven at least in part by gut-origin inflammation. |
Landmark research demonstrated that the gut microbiome of obese mice could transfer metabolic dysfunction to lean germ-free mice through transplantation. Mice receiving microbiomes from obese donors gained significantly more fat than those receiving microbiomes from lean donors — despite identical food intake. The microbiome composition itself was the independent variable driving fat accumulation. (Turnbaugh PJ et al. — Nature, 2006)
The implication is significant: metabolic dysfunction can be transmitted through the microbiome independently of diet. The composition of gut bacteria is not a side effect of poor metabolic health — it is a driver of it. And because the cascade is self-reinforcing — insulin resistance further disrupts the microbiome, which further worsens insulin resistance — the intervention has to address the gut alongside the diet.

Short-Chain Fatty Acids — The Metabolic Output Most People Have Never Heard Of
Understanding short-chain fatty acids was the moment gut health stopped being abstract for me and became something I could actually work with. These are not incidental byproducts. They are the mechanism — the specific compounds your gut bacteria produce when you feed them properly, and the specific thing that goes missing when your microbiome is disrupted.
SCFAs — primarily butyrate, propionate, and acetate — are produced when beneficial gut bacteria ferment dietary fiber. Without adequate fiber, the bacteria that make them starve. Without adequate butyrate, the cells lining the colon lose their primary fuel source, the gut barrier weakens, and the inflammatory cascade that drives insulin resistance begins.
Butyrate — directly improves insulin sensitivity in muscle and liver cells through HDAC inhibition — epigenetic regulation of genes involved in glucose metabolism — and serves as the primary fuel for colonocytes, the cells lining the colon
Propionate — is transported to the liver where it reduces fat production and improves glucose regulation
Acetate — circulates systemically and influences appetite regulation, energy expenditure, and fat storage
In people with a low-diversity microbiome — the pattern consistently seen in insulin resistance, obesity, and type 2 diabetes — SCFA production is substantially reduced. Less butyrate means a weaker gut barrier, more systemic inflammation, and impaired insulin sensitivity. The downstream effects of losing this one output ripple through the entire metabolic system.
"Your gut bacteria are producing compounds that directly improve insulin sensitivity. The question is whether you have enough of the right bacteria — and whether you're feeding them."
What Destroys Gut Microbiome Diversity
The modern gut microbiome is substantially less diverse than that of pre-industrial populations. Research comparing gut microbiomes across cultures found that populations with traditional whole-food diets had two to three times the microbial diversity of Western populations — and dramatically lower rates of metabolic disease. Four factors most consistently associated with reduced diversity:
1. Ultra-processed foods — fiber-poor, additive-rich, and designed for preservation and palatability — not microbial health. They starve beneficial bacteria while feeding pathogenic species. Emulsifiers in particular — including carboxymethylcellulose and polysorbate-80 — directly disrupt the gut barrier and alter microbiome composition toward inflammatory species. As covered in the Ultra-Processed Food article in this library, this damage operates independently of calories.
2. Antibiotics — a single course of broad-spectrum antibiotics can reduce gut microbiome diversity by 25–50%. Some diversity recovers within weeks; some species do not return for months or years, and in some individuals, never fully recover. Repeated antibiotic exposure produces cumulative diversity loss with long-term metabolic consequences. This does not mean avoiding necessary antibiotics — it means understanding the recovery work that follows.
3. Chronic stress and poor sleep — the gut-brain axis runs bidirectionally. Chronic psychological stress and sleep disruption alter gut motility, intestinal permeability, and microbiome composition through cortisol and autonomic nervous system signaling. Elevated cortisol directly reduces populations of Lactobacillus and Bifidobacterium — two of the most important beneficial genera. As covered in the Sleep Metabolism article in this library, the sleep-gut-metabolism connection is underappreciated.
4. Low dietary fiber — dietary fiber is the primary food source for beneficial gut bacteria. The average American consumes 10 to 15 grams of fiber per day against a recommended minimum of 25 to 38 grams. This chronic underfeeding starves the species that produce butyrate and other SCFAs, allowing less beneficial species to dominate by default.
A randomized controlled trial comparing high-fiber diets to high-fermented food diets found that high-fermented food intake significantly increased microbiome diversity and reduced 19 inflammatory proteins measured in blood samples. High-fiber diets increased SCFA output but did not consistently increase diversity — suggesting that fiber and fermented foods address different aspects of microbiome health and both are needed.
(Sonnenburg JL et al. — Cell, 2021)

2–3×
greater gut microbiome diversity in populations eating traditional whole-food diets compared to Western populations — and dramatically lower rates of metabolic disease
Zmora N et al. — Nature Reviews Gastroenterology & Hepatology, 2019
How to Improve Your Gut Microbiome Naturally — What the Evidence Supports
Microbiome recovery is not a rapid process. Significant changes in microbial composition take weeks to months, and some diversity loss from early life exposures may never fully reverse. But the research on what drives improvement is clear — and the interventions overlap almost entirely with the MAP30 dietary framework, which is not a coincidence. Here is what the evidence consistently supports:
1. Dietary fiber — the non-negotiable foundation — increasing dietary fiber from diverse plant sources is the most consistently supported intervention for increasing SCFA production and gut microbiome function. The target is not a specific food but diversity of fiber sources — different fibers feed different bacterial species. Eating 30 different plant foods per week has been associated with significantly higher gut diversity in large population studies. Non-starchy vegetables, legumes, and whole intact foods are the primary sources in a low-carbohydrate framework.
2. Fermented foods — for diversity, not just probiotics — the Sonnenburg trial established that fermented foods — yogurt, kefir, kimchi, sauerkraut, kombucha — increase gut microbiome diversity more reliably than fiber alone, and do so by introducing live microbial communities rather than just feeding existing ones. Daily consumption of fermented foods is one of the most evidence-supported practical interventions for microbiome health. I made fermented foods a daily part of my own diet and noticed the difference in digestive consistency within three to four weeks.
3. Fasting — the microbiome reset mechanism — fasting periods allow the gut lining to repair, reduce intestinal inflammation, and shift the microbiome toward more beneficial populations. Research on intermittent fasting specifically has found increases in Akkermansia muciniphila — a species strongly associated with gut barrier integrity and improved metabolic function — after fasting interventions. As covered in the Fasting articles in this library, the fasting window creates the low-insulin, low-inflammation environment in which gut barrier repair occurs most effectively.
4. Eliminating ultra-processed foods — no microbiome intervention produces lasting improvement in the presence of a diet rich in ultra-processed foods. The emulsifiers, artificial sweeteners, preservatives, and low-fiber structure of ultra-processed foods continuously degrade microbiome diversity. Removing them is not a dietary preference — it is a precondition for gut recovery.

One specific note on artificial sweeteners: multiple studies, including a 2022 randomized controlled trial in Cell, found that saccharin, sucralose, and other non-caloric sweeteners alter gut microbiome composition and impair glucose tolerance in healthy adults. They are not metabolically inert. The Qin et al. metagenome study found that type 2 diabetes was associated with a specific pattern of gut dysbiosis — reduced butyrate-producing bacteria, increased opportunistic pathogens — independent of conventional risk factors. The microbiome was a predictor of metabolic status, not just a consequence of it.
The gut-insulin resistance connection is the thread that runs through the entire MAP30 framework. If you have been following a clean diet and still experiencing persistent fatigue, brain fog, irregular digestion, or stubborn metabolic markers, the gut may be the variable that has not yet been addressed. Metabolic recovery without attention to the gut is recovery built on an unstable foundation.
This article is part of our Gut Health & Recovery series.
Link Here: Gut Health & Recovery
Latest Articles

Why Is Sugar So Addictive
Sugar addiction is not a character flaw or a lack of willpower. It is a neurochemical process — one the food industry spent decades studying, and exploiting.
What Are Hormones
Most people think about weight gain as a math problem — calories in versus calories out. The biology is more complex than that. Your body is run by a hormonal system.

Ultra-Processed Food
Ultra-processed food is not simply unhealthy food. It is an industrially manufactured product engineered to override hunger and drive compulsive consumption.

John Shaw
MAP30 Challenge
John Shaw is a Certified Nutrition Educator and the founder of the MAP30 Challenge. What began as a personal health journey at 294 pounds, and pre-diabetic, evolved into a structured 30-day metabolic reset program grounded in nutritional science. John's mission is simple: give people the biological education that the diet industry never did.
FAQ's
1. How do you improve gut microbiome naturally?
The most consistently evidence-supported interventions are: increasing dietary fiber from diverse plant sources (30 different plant foods per week is an evidence-backed target); daily consumption of fermented foods such as yogurt, kefir, kimchi, and sauerkraut; eliminating ultra-processed foods and emulsifiers; incorporating fasting periods to allow gut barrier repair; and avoiding unnecessary antibiotic use. Significant microbiome changes take weeks to months. No single intervention is sufficient — the combination produces results that individual interventions do not.
2. What does the gut microbiome do?
The gut microbiome ferments dietary fiber into short-chain fatty acids that fuel intestinal cells and regulate blood sugar; trains the immune system; produces over 90% of the body's serotonin; maintains the gut barrier; and modulates metabolic hormones controlling appetite and glucose metabolism. It is a primary metabolic organ, not a secondary digestive system. Disrupting it disrupts all of these functions simultaneously.
3. How does gut health affect insulin resistance?
A disrupted microbiome produces less butyrate, which weakens the gut barrier. Bacterial fragments enter the bloodstream, triggering systemic inflammation. Inflammatory cytokines — particularly TNF-alpha and IL-6 — directly impair insulin signaling in muscle and liver cells, driving insulin resistance. This cascade has been documented across multiple clinical study designs and operates independently of diet — meaning gut-driven inflammation can maintain insulin resistance even when dietary improvements have been made.
4. What are short-chain fatty acids and why do they matter?
Short-chain fatty acids — primarily butyrate, propionate, and acetate — are produced when beneficial gut bacteria ferment dietary fiber. Butyrate is the primary fuel for colon cells and improves insulin sensitivity through epigenetic mechanisms. Propionate reduces liver fat production. Acetate influences appetite and energy metabolism. In low-diversity microbiomes — the pattern seen in insulin resistance and type 2 diabetes — SCFA production is substantially reduced, with downstream effects across metabolic function.
5. What is leaky gut and is it real?
Increased intestinal permeability — commonly called leaky gut — refers to a breakdown in the tight junctions between intestinal cells, allowing bacterial fragments and undigested molecules to enter the bloodstream. The underlying mechanism is well-documented in research, measurable, associated with metabolic dysfunction, and responsive to dietary intervention. The term is contested in mainstream medicine but the biology is not — lipopolysaccharide translocation from the gut into the portal circulation is a documented driver of the systemic inflammation that underlies metabolic syndrome.
6. Do artificial sweeteners affect the gut microbiome?
Yes. A 2022 randomized controlled trial published in Cell found that saccharin, sucralose, and other non-caloric sweeteners alter gut microbiome composition and impair glucose tolerance in healthy adults. They are not metabolically inert. People managing insulin resistance or metabolic syndrome should approach artificial sweeteners with the same caution applied to added sugar — they change the microbiome in ways that undermine the same metabolic recovery that dietary changes are trying to produce.
Sources
Turnbaugh PJ et al. — 'An obesity-associated gut microbiome with increased capacity for energy harvest' (Nature, 2006) — https://pubmed.ncbi.nlm.nih.gov/17183312/
Sonnenburg JL et al. — 'Gut-microbiota-targeted diets modulate human immune status' (Cell, 2021) — https://pubmed.ncbi.nlm.nih.gov/34256014/
Qin J et al. — 'A metagenome-wide association study of gut microbiota in type 2 diabetes' (Nature, 2012) — https://pubmed.ncbi.nlm.nih.gov/22797518/
Suez J et al. — 'Personalized microbiome-induced adiposity involves sucralose- and saccharin-mediated changes in gut microbiota' (Cell, 2022) — https://pubmed.ncbi.nlm.nih.gov/35907413/
Cani PD et al. — 'Metabolic Endotoxemia Initiates Obesity and Insulin Resistance' (Diabetes, 2007) — https://pubmed.ncbi.nlm.nih.gov/17519319/
Zmora N et al. — 'You are what you eat: diet, health and the gut microbiota' (Nature Reviews Gastroenterology & Hepatology, 2019) — https://pubmed.ncbi.nlm.nih.gov/30262889/

Let Me See The Ai Audit Report
I had the MAP30 course material audited and graded by 4 dirrerent Ai models. I was shocked by the grade. See what they had to say about the MAP30 course.

Jaabsha Marketing LLC | All rights reserved 2024 Terms Of Service
